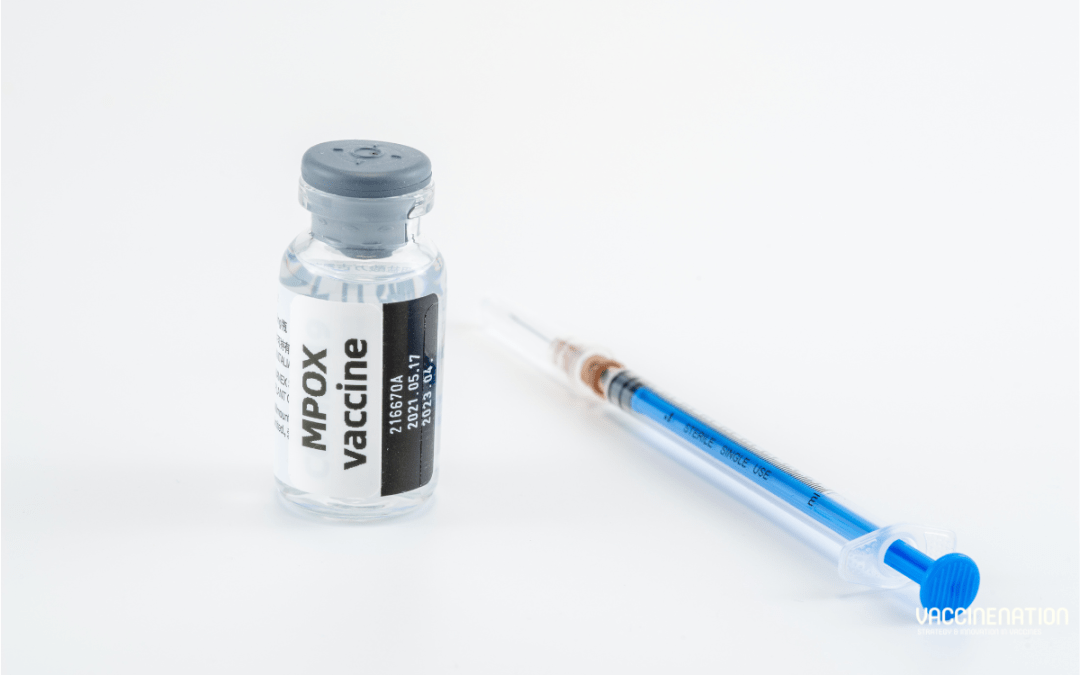by Charlotte Kilpatrick | Oct 24, 2024 | Technology |
The University of Connecticut (UConn) announced in October 2024 that associate Professor Thanh Nguyen’s research has received “significant” backing from The Bill and Melinda Gates Foundation. The Gates Foundation has awarded a series of grants totalling $6.6 million, following support from the National Institutes of Health (NIH) and the US Department of Agriculture (USDA). The funding will contribute to research and innovation for a microneedle array patch that can deliver multiple human vaccines at once. The Foundation initially awarded $2 million, which has increased after early success.
Microneedle array patch technology
Dr Thanh Nguyen works in the College of Engineering’s School of Mechanical, Aerospace, and Manufacturing Engineering. His microneedle method is “far less painful” than traditional syringe delivery and offers access and uptake benefits.
“What if we were able to mail people vaccines that don’t need refrigeration, and they could apply to their own skin like a bandage?”
The technology delivers highly concentrated vaccines in powder from over months, through a “nearly painless” 1-centimetre-square biodegradable patch.
“The primary argument is that getting vaccines and boosters is a pain. You have to go back two or three times to get these shots. With the microneedle platform, you put it on once, and it’s done.”
Funding increases
After the initial award of $2 million, the project made good progress and received additional funding to support the development of a scale-up manufacturing technology to produce patches on an industrial scale. In late September, the Gates Foundation awarded $4 million to take the patch “a step farther” as a pentavalent and Polio vaccine targeting diphtheria, tetanus, pertussis, HIV, Hepatitis B, and Polio. With this funding, the team can “build up productivity”. They are partnering with LTS to scale up production and are expanding the size of laboratory.
The award also marks a fundraising milestone for Dr Nguyen, who has earned more than $25 million in research awards, which he reflects “doesn’t come naturally”.
“It comes from the recognition of the high impact of the research and the lab’s success in publishing articles. It is a testament to the importance of what we are doing.”
For the latest vaccine development and technology updates, don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 23, 2024 | Global Health |
A study in The Lancet Global Health sought to provide counterfactual scenarios to evaluate the short-term effects of different vaccination strategies on mpox cases and deaths in the Democratic Republic of the Congo (DRC). The researchers used a dynamic transmission model to simulate mpox transmission, stratified by age and province; this was used to assess potential vaccination strategies and their effects on deaths and cases in an epidemic year. The results indicate that vaccinating children aged 15 years or younger, or younger than 5 years, in endemic regions, would be the “most efficient use of vaccines” when resources are limited.
Mpox in DRC
Mpox was first identified in the Democratic Republic of the Congo (DRC) in 1970; it is a zoonotic infectious disease caused by the monkeypox virus (MPXV), which is endemic in “numerous regions” of west and central Africa. MPXV has two clades:
- Clade I is endemic in central Africa with an estimated case fatality rate of up to 10% and mainly affecting children. It is divided into two subclades, Ia and Ib.
- Clade II was historically found in west Africa, with an estimated case fatality rate of up to 1%-3%. It is also divided into two subclades, IIa and IIb. Clade IIb was responsible for the global mpox epidemic in 2022.
The authors state that, until 2022, MPXV was not associated with large outbreaks; most cases were related directly to sylvatic transmission from animals to humans via hunting, wild game preparation, and consumption. Increases in human-to-human transmission were identified in 2017.
The researchers suggest that the low likelihood of transmission in the early decades after the virus’ discovery could be related to smallpox eradication programmes, which offered cross-immunity via vaccination against a related orthopoxvirus. Indeed, since the cessation of the smallpox vaccination programme in the DRC, there has been a “concurrent increase in mpox cases and outbreak frequency”. There is an ongoing, “unprecedentedly large” outbreak of clade I mpox in the DRC, with more than 14,000 reported suspected cases by the end of 2023 and a 4.6% case fatality rate. Over 70% of the deaths are in children younger than 15 years.
Genetic analyses of clade Ia MPXV genomes indicate that in this outbreak, multiple, independent zoonotic introductions into the human population have occurred from one or more reservoir species. An increasing burden of clade Ib MPXV infections have been identified in eastern DRC with evidence of “sustained” human-to-human transmission and many cases in women aged 15-29 years, but clade Ia infections continue to comprise most mpox cases in the DRC.
The study
Bavarian Nordic’s modified vaccinia Ankara vaccine (JYNNEOS) is protective against mpox. It was approved by the US FDA in 2019 but was not widely used against mpox until the 2022 outbreak, when it was “quickly mobilised to vaccinate people at high risk of infection in the USA and Europe”. Despite its high efficacy at two doses, it is “largely unavailable” outside the USA and Europe.
The authors aimed to inform policy and decision makers on the “potential benefits of, and resources needed,” for mpox vaccination campaigns in the DRC. They used an approach based on models from operations research and decision science to offer a robust analysis of policy choices “even in the context of incomplete and uncertain data”. The study uses mathematical modelling to simulate the spread of mpox in the DRC during 2023.
Without vaccination, the model predicted 14,700 cases of mpox and 700 deaths from mpox in the DRC over 365 days, consistent with reported estimates. Almost 50% of the cases and deaths came from the province of Equateur. Cases were evenly split between the three age groups: 34% in children under 5 years, 32% in children aged 5-15 years, and 34% in people older than 15 years. However, deaths were “predominantly” seen in children younger than 5 years (51%).
Vaccinating 80% of children younger than 5 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 10,500 cases and 400 deaths. Vaccinating in endemic provinces increased cases to 10,700 and deaths remained the same. The numbers of vaccine doses needed for the strategies were 41.4 million (all provinces), 33.8 million (provinces with a history of mpox), and 13.2 million (endemic provinces only).
Vaccinating 80% of children younger than 15 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 6,400 cases and 200 deaths. Vaccinating in endemic provinces increased cases to 6,800 and deaths remained the same. The numbers of vaccine doses required for these strategies were 81.6 million (all provinces), 67.1 million (provinces with a history of mpox), and 26.6 million (endemic provinces only).
Vaccinating 80% of all ages in all provinces or only non-endemic provinces with a history of cases decreased the case burden to 1,400 cases and 100 deaths, and 2,000 cases and 100 deaths when vaccinating in provinces endemic for mpox. The numbers of doses required for these strategies were 170.8 million (all provinces), 142.0 million (provinces with a history of mpox), and 56.8 million (endemic provinces only).
Managing resources
The paper finds that vaccinating all ages leads to the “largest impact on magnitude of cases and deaths”, but that vaccinating only children aged 15 years or younger provides “nearly the same effect with fewer vaccine doses required”. Although vaccinating only children younger than 5 years showed a “drop-off” in averted cases and deaths, it provides the most efficiency.
“This analysis shows the effectiveness of focussing an mpox vaccination campaign specifically in the provinces endemic for mpox in the DRC. This targeted strategy prevents nearly as many cases and deaths as broader approaches but uses fewer vaccine doses and thus would be less costly to implement.”
Alexandra Savinkina, fourth year PhD student in the Yale School of Public Health (YSPH) Department of Epidemiology (Microbial Diseases), commented that this study could influence vaccination policy.
“My hope is that it could help inform policy for vaccination in the country and potentially the region and move the needle forward on getting vaccines to the people who need them most in the DRC.”
Savinkina hopes that “we can learn from the global mpox outbreak that we can’t ignore disease in other places”.
“If the resources to help people exist, I think we should be using them, whether in the U.S. or in Africa.”
Dr Gregg Gonsalves, associate professor of epidemiology at YSPH, acknowledged barriers to access.
“We take it for granted that we can get a vaccination for COVID or a flu shot at our local CVS, but the infrastructure to deliver vaccines in DRC is far less robust.”
For more vaccine research updates, subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 17, 2024 | Global Health |
A study in The Lancet Global Health estimates the impact that the 100 Days Mission could have had on the COVID-19 pandemic. The authors find that the implementation of non-pharmaceutical interventions (NPIs) within the 100 Days Mission could have averted around 8.33 million deaths, corresponding to a monetary saving of US$14.35 trillion. Investment in manufacturing and health systems contribute an increase to 11.01 million deaths averted. The researchers comment on the value of the 100 Days Mission but emphasise the importance of “prioritising a more equitable global vaccine distribution”.
In search of greater vaccine benefits
Although COVID-19 vaccines are estimated to have prevented almost 20 million deaths, the authors demand a better understanding of the “further health and economic benefits that could have been achieved” through shorter development times and “improved global equity in pandemic preparedness”. CEPI’s 100 Days Mission was proposed in 2021, establishing the ambition of cutting vaccine development time for new pathogens to 100 days. This about a third of the time taken to deliver the first COVID-19 vaccine.
“The availability of COVID-19 vaccines within 100 days would have substantially changed the pandemic; however, these benefits would be finite without enabling equitable access to vaccine products through system equity.”
Various efforts to encourage global vaccine distribution were “hampered” by inequities, and it is recognised that manufacturing capacity should be “expanded but also diversified” to “promote self-sufficiency and regional resilience”. Furthermore, supply chains and infrastructure must be scaled to enable the delivery of vaccines that rely on cold-chain infrastructure.
The study
The searchers hoped to quantify the potential impact of the 100 Days Mission by retrospectively estimating the effect it would have had on the COVID-19 pandemic, thus offering evidence to support decision making around future investments in research and development capabilities. They also aimed to quantify the impact of “additional investments”.
The analyses use an extended version of a previously published compartmental susceptible-exposed-infectious-recovered transmission model of COVID-19 vaccination with an explicit healthcare pathway. The vaccination pathway was expanded to include booster vaccination alongside waning efficacy, capturing the “restoration of immunity” through booster doses. The new vaccination pathway was parameterised to match platform-specific vaccine efficacy data and the duration of protection.
The authors modelled the impact of the 100 Days Mission by simulating a counterfactual scenario where the global vaccination campaign began on 20th April 2020, 100 days after the publication of the full SARS-CoV-2 genome. This scenario assumes that vaccinations in each country took the same roll-out process, but 232 days earlier. Two additional scenarios reflected “increased investment” in research and delivery infrastructure.
The Manufacturing scenario removed supply constraints, enabling the rollout of vaccination on 20th April 2020 in every country, without stockouts. The infrastructure-equity scenario enhanced both national health systems and global distribution networks so that all countries achieved 40% vaccine coverage in the first year and 40% booster coverage in the second year.
To account for the relaxing of non-pharmaceutical interventions (NPIs), the authors simulated three scenarios for NPI relaxation speeds as vaccination coverage improved. The History scenario assumed no changes, whereas the Target and Economic scenarios assumed earlier relaxing; the Target scenario lifted all restrictions over two months after reaching more than 80% adult coverage in high-income countries or more than 80% coverage in those older than 60 in other countries. The Economic scenario lifted NPIs more gradually after reaching the over-60 target, prioritising the reopening of schools.
Study findings
The results suggest that the 100 Days Mission could have averted an additional 8.33 million deaths due to COVID-19 by the end of 2021 when combined with the History NPI lifting scenario. In this scenario, an estimated 26.72 million severe cases of COVID-19 requiring hospitalisation and 1/44 billion infections would have been averted. Most of these averted deaths, hospitalisations, and infections would have occurred in low- and middle-income countries (LMICs).
The estimated VSLs (value of a statistical life) that could have been saved by the 100 Days Mission through the History scenario is US$22.61 trillion globally. As VSLs are “significantly higher” in HICs, 57% of the global value of statistical life averted occurred in HICs, even though most deaths would have been averted in LMICs. To estimate the monetary values associated with lives saved the authors multiplied the number of lives saved by the country-specific monetary VSL and by the value of a statistical life-year (VSLY).
“Increased investment in both global manufacturing and health systems infrastructure further increases the number of deaths that could be averted and the associated health-economic savings.”
In the 100 Days Mission with both manufacturing and health systems investments, an estimated 11.01 million deaths could have been averted and a value of $31.29 trillion in statistical lives saved. However, the authors describe this scenario as “unlikely”. In all scenarios involving the relaxing of NPIs thanks to earlier availability of vaccines, additional lives would have been saved.
In the Target NPI lifting scenario, an estimated 5.76 million deaths (100 Days Mission alone) to 9.20 million deaths (100 Days Mission with both manufacturing and infrastructure investments) could have been averted. In these scenarios, 12,600 and 23,900 fewer days of NPIs would have been implemented globally: 70 days and 133 days on average per country. Under the Economic scenario there were “similar trade-offs between public health and economic gains”. The public health and health economic outcomes would be greater than under the Target scenario, but still lower than the History scenarios.
Substantial benefits
“Earlier access to COVID-19 vaccines could have had substantial benefits.”
Most of the estimated averted deaths would have been concentrated in LMICs, but this demands investments in vaccine research, supported by “improvements to manufacturing and health system infrastructures”. With these investments, the authors estimate that 11 million deaths could have been prevented globally.
Although NPIs were effective at reducing transmission they incurred “significant economic and societal costs”, including consequences for education. Therefore, a major benefit of earlier access to vaccination is the reduction in school closures; in the Economic scenario, prioritising school opening could have averted 1,120 weeks of full school closures and 2,490 of partial school closures. This represents an average of 6 weeks of fully open schools and 14 weeks of partly open schools per country.
“Reopening schools and relaxing NPIs safely will crucially require scaling up both vaccine delivery infrastructure and manufacturing. Without addressing both aspects, advancements in vaccine development speed might not translate into equitable benefits globally.”
The results emphasise the importance of investments in support of the 100 Days Mission in controlling a future potential pandemic, with benefits for both health and economy.
“The 100 Days Mission is ambitious, requiring global innovation through creating vaccine libraries, clinical trial networks, accelerated immune response marker identification, rapid vaccine manufacturing, and strengthened global disease surveillance.”
CEO of CEPI, Dr Richard Hatchett, hopes that this research will encourage global commitment to the 100 Days Mission.
“This work shows in the starkest terms why the world needs to be prepared to move faster and more equitably when novel pandemic disease threats emerge. Investing in preparedness now to make the 100 Days Mission possible for future incipient pandemics will save millions upon millions of lives and protect the global economy against catastrophic losses.”
Join us at the Congress in Barcelona this month to explore how lessons from the COVID-19 pandemic can inform better global preparedness for pandemic and epidemic pressures, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 16, 2024 | Global Health |
In October 2024, parliamentarians from across the world met at the UNITE Global Summit in Berlin to sign a statement in support of the WHO Pandemic Agreement. The statement was described by WHO as a “significant commitment” to “strengthen pandemic preparedness, response, and equitable access to health”. The Summit was held in collaboration with the World Health Summit (WHS), drawing global parliamentarians, civil society leaders, and health experts to “translate discussions into actionable policy priorities”.
Building trust for a healthier world
The World Health Summit 2024 was held under the theme “building trust for a healthier world”, focussing on inspiring “innovative solutions for better health and well-being for all”. The UNITE Global Summit agenda reflected key topics highlighted during WHS, divided into four pillars:
- Human rights and equitable access to health
- Global health architecture and security
- Strengthening of healthcare systems
- Sustainable financing for health
Support for the Pandemic Agreement
The Pandemic Agreement is under negotiation by WHO Member States as an attempt to address the “gaps” exposed by the COVID-19 pandemic and the threats presented by mpox and other disease outbreaks. It also seeks to “strengthen global collaboration pandemic prevention, preparedness, and response”.
A critical moment during the summit was the signing of the Global Parliamentary Statement in Support of the Pandemic Agreement, which demonstrated the importance of parliamentarians in “ensuring global health security and safeguarding populations against future pandemics”. The statement emphasised their commitment to ensuring that “all countries, regardless of resources, have access to the tools, capacities, resources, and healthcare required” during a pandemic.
Dr Ricardo Baptista Leite, President of UNITE, commented that parliamentarians are the “voice of the people” and carry a “crucial responsibility in safeguarding public health”.
“The WHO Pandemic Agreement represents a historic opportunity to prevent pandemics and strengthen our global preparedness and response capabilities. By signing this statement, we are not only showing our support for the agreement but also pledging to ensure that its principles of equity, solidarity, and global cooperation are fully realised in every nation.”
WHO Director-General Dr Tedros Adhanom Ghebreyesus welcomed the support for the “once-in-a-generation opportunity to build a stronger, fairer, and more prepared global health system”.
“By signing this statement, parliamentarians from around the world are showing their commitment to protecting lives from future pandemics and ensuring equitable access to vaccines, treatments, diagnostics, and other health tools for every country, particularly those with fewer resources.”
Key commitments
The Global Parliamentary Statement emphasises four “key commitments”:
- Equity at the core – ensuring equitable access to pandemic-related health tools based on public health need for everyone, especially countries with fewer resources
- Global solidarity – strengthening international cooperation to build resilient health systems that can prevent and respond to pandemics
- Legislative action – advocating for the ratification and implementation of the Pandemic Agreement within national legislatures, as appropriate
- Combating misinformation – providing communities with evidence-based health information to counter the spread of harmful misinformation
Dr Baptista Leite reflected that “the challenges we face today demand a global response”.
“No single country can prevent or combat pandemics alone. The WHO Pandemic Agreement is an essential step forward in ensuring that every nation has the tools, resources, and capabilities to respond to future health threats.”
More work to be done
WHO states that the Pandemic Agreement “needs to continue to garner broad international support”. Within the latest commitment, parliamentarians are to work with WHO and other international organisations to ensure the Pandemic Agreement is implemented in a way that “benefits all countries”, particularly those with “limited resources”.
“The statement signed in Berlin is expected to act as a catalyst for global parliamentary action, fostering collaboration and solidarity among nations.”
Join us at the Congress in Barcelona this month for a keynote discussion on “strengthening global preparedness and building resilient health systems” with experts from the field, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 16, 2024 | Technology |
CEPI announced in October 2024 that it is working with experts at the National Research Council of Canada (NRC) to bioengineer a “commonly used approach” to safely make protein antigens in “as little as two weeks”. This would be between eight and twelve times faster than the current timeline of antigen production for protein-based vaccines. CEPI is contributing up to CAD $850,000 and the NRC is providing up to CAD $308,000 (in kind) to establish proof-of-technology.
Low cost and high speed
CEPI notes the importance of manufacturers being able to make “sufficient quantities” of vaccine components at low cost to enable mass production. Although mammalian cell lines are a common choice for vaccine processes, boasting ease of culture and a high production yield, they can take four to six months to develop and optimise for antigen production. This is a “major challenge” to efforts to develop vaccines quickly in response to fast-spreading viral outbreaks.
An optimised approach
Scientists at the NRC have developed a mammalian cell line that could be optimised for rapid antigen production. The research is expected to “majorly accelerate” the time needed for infectious disease vaccine development, says CEPI’s Executive Director of Manufacturing and Supply Chain (Acting), Ingrid Kromann.
“If successful, this optimised cell line could help vaccine doses be more rapidly available for clinical trials and initial emergency use during future outbreaks, supporting CEPI’s goal – embraced by Canada, and other G7 and G20 nations – to respond to a novel virus with a new vaccine in just 100 days after its discovery.”
Importantly, the technology is going to be suitable for transfer to low- and middle-income countries, enabling “local and rapid” vaccine production closer to the source of a future outbreak and improving accessibility. Dr Lakshmi Krishnan, Vice President of Life Sciences at the NRC, looks forward to working with CEPI to take the platform technologies forward to “accessible tools that could help accelerate vaccine production around the world”.
“Recognising the critical need for rapid vaccine production during a health emergency, this research and development project in our labs will advance innovative technologies to improve biomanufacturing processes and increase the efficiency of large-scale manufacturing of vaccines and other biologics.”
For the latest in vaccine technology for improved accessibility, join us at the Congress in Barcelona this month, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 14, 2024 | Global Health |
In October 2024 the European Investment Bank (EIB Global) announced €2 million financing for early-stage vaccine development in Rwanda by Akagera Medicines Africa Limited. The support is intended to accelerate research, development, and manufacturing of new vaccines against infectious diseases like tuberculosis, HIV, Lassa fever, and Ebola. It will also be used to “strengthen technical skills and expertise” to support “home-grown discovery, manufacturing, and development of vaccine delivery systems” in Rwanda.
Global Gateway
This financing is part of the EU Global Gateway initiative, a strategy to “boost smart, clean, and secure links in digital, energy, and transport sectors and to strengthen health, education, and research systems”. Team Europe is mobilising up to €300 million between 2021 and 2027 to “allow EU’s partners to develop their societies and economies” whilst creating opportunities for EU Member States to “invest and remain competitive”. EIB Global supports “high impact investment” to enhance healthcare and pharmaceutical manufacturing, encourage greater “health resilience”, and support equitable access to healthcare.
Continent-based solutions
EIB Global states that Africa bears the highest disease burden globally, demanding “more home-grown or continent-based solutions”.
“Vaccination is a critical activity to ensure and guide investments in universal health and has a crucial role to play in achieving 14 of the 17 United Nations Sustainable Development Goals.”
Akagera Medicines was founded in 2018 and registered a 100%-owned subsidiary in Kigali in 2022. Its mission is “targeting tuberculosis and other infectious diseases with liposomal nanotherapeutics”. Commenting on the financing announcement at the World Health Summit in Berlin, Chief Executive Officer Michael Fairbanks recognised the “significant support” of the European Investment Bank.
“We are now a clinical company and moving faster to build human capacity and specialised infrastructure in Africa to support vaccine development.”
CEO of the Rwanda Social Security Board (RSSB) Regis Rugemanshuro stated that the financial support is an “important contribution to the realisation of Rwanda’s vision to become a biotech hub” and the wider vision of “Africa becoming self-reliant in vaccine and medicine manufacturing”.
“RSSB is looking forward to deepening partnerships with EIB and other international institutions to build resilient healthcare ecosystems in Rwanda and in Africa.”
Vice President of EIB Thomas Ostros identified the Bank’s “close cooperation with public and private partners” to “accelerate development of innovative solutions”.
“The EIB is committed to further strengthening our partnership with local and international players, to scale up investment and support innovative technology together.”
Belen Calvo Uyarra, EU Ambassador to Rwanda, agreed that the investment was another “important milestone”.
“Through Global Gateway, the EU is focussed on advancing equitable access to health products and local manufacturing in Africa.”
For more from key players in efforts to establish local manufacturing capacities in Africa and champions of equitable access to health products, join us at the Congress in Barcelona later this month. Don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 14, 2024 | Technology |
SK bioscience announced in October 2024 that it has signed an agreement to acquire a stake in Fina Biosolutions (FinaBio) with a $3 million investment. SK bioscience becomes FinaBio’s first and sole strategic investor with a goal of improving the immunogenicity and productivity of conjugate vaccines. This announcement is another example of SK bioscience’s investment in global companies to “create synergies in business” after recently completing the acquisition of a controlling stake in IDT Biologika. The company states that it is securing its “competitiveness” through strategic investments in “promising companies with exceptional technology” and M&As to “lay the foundation for a great leap forward into a leading global company”.
FinaBio’s technology
Founded in 2006, FinaBio seeks to “help emerging market vaccine manufacturers learn to make affordable protein polysaccharide conjugates for vaccines”. It is now a “premier provider” of laboratory and consulting services, specialising in the research and development of conjugate vaccines for pneumoniae, meningococcal, typhoid, and other diseases. One of FinaBio’s key assets is FinaXpress, a proprietary E. coli expression system, that can produce proteins not previously made in the bacteria, like the carrier protein CRM197. FinaBio has expanded access to this protein, marketed as EcoCRM.
FinaBio is also developing a next-generation conjugation technology that is site-specific and targets the desired location for antigen binding. This is intended to boost immunogenicity and productivity. Supplying conjugation technology and carrier proteins to various global biotech companies and institutions, FinaBio continues to expand its business units.
A conjugate collaboration
SK bioscience will use FinaBio’s CRM197 technology in its efforts to “secure the high effectiveness of diverse conjugate vaccines while increasing profitability through high-yield processes”. CEO and President of SK bioscience Jaeyong Ahn is “delighted to continue developing partnerships with global firms that have next-generation vaccine technology”.
“Through our mid- to long-term collaboration with FinaBio, we will advance the vaccines we are developing to the next level and strengthen our competitiveness for global market expansion.”
Dr Andrew Lees, Founder and CEO of FinaBio, apprecitaes SK’s “confidence” in the organisation and support of accelerated global commercialisation of EcoCRM.
“Combined with our efficient conjugation technology, this will enable the development of the next generation conjugate vaccines. It will also allow us to continue our mission of promoting affordable vaccines.”
We look forward to welcoming FinaBio back to the exhibition floor at the Congress in Barcelona later this month; get your tickets to connect with their team there and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 10, 2024 | Global Health |
The African Union Development Agency – New Partnership for Africa’s Development (AUDA-NEPAD) announced in October 2024 that Wellcome is granting US$12,301,075 to “support the strengthening and harmonisation of regulatory systems and the operationalisation” of the African Medicines Agency (AMA). The grant will help to drive efforts to “overcome regulatory capacity challenges” to improve access to essential medical products and technologies.
The African Medicines Regulatory Harmonisation (AMRH) initiative has been “pivotal” at creating a “cohesive regulatory environment” for the pharmaceutical sector across Africa since 2009. It focuses on using Regional Economic Communities (RECs) to ensure that African populations have access to high-quality, safe, and effective medical products and health technologies. Wellcome’s grant will be used in alignment with AMRH’s vision of “overcoming barriers” like “limited human and institutional capacity, fragmented regulatory processes, and inconsistent technical standards”.
AMA
AMA’s vision is that “African people have access to essential medical products and technologies”; it hopes to achieve this through the mission: “provide leadership in creating an enabling regulatory environment for pharmaceutical sector development in Africa”. The funding is expected to accelerate efforts to create a “unified and efficient” regulatory framework. The partnership between Wellcome and AUDA-NEPAD is a “major advancement in the pursuit of a robust and harmonised regulatory environment” with positive effects for health outcomes in Africa.
Efficient, connected, fair
Symerre Grey-Johnson Director for Human Capital and Institutional Development at AUDA-NEPAD, stated that the “generous support” from Wellcome is a “crucial endorsement of our mission”.
“With the African Medicines Regulatory Harmonisation (AMRH) intiative laying the groundwork for the African Medicines Agency (AMA), this grant will empower us to address significant regulatory challenges and enhance access to essential medical products for millions of Africans.”
Mr Grey-Johnson believes that the collaboration will “solidify the foundation of the AMA” and ensure a “robust and harmonised” regulatory environment across the continent. Dr Sally Nicholas, Wellcome’s Head of Health Systems and Environment, recognised the AMA’s “crucial role” in creating a “more efficient, connected, and fair regulatory system” in Africa.
“Strengthening regulatory systems is fundamental to improving healthcare outcomes for Africa. By supporting innovative partnerships, initiatives, and solutions to help coordinate effectively operationalise the AMA, we can ensure equitable access to much-needed vaccines, treatments, and interventions for those with the greatest need.”
At the Congress in Barcelona this month we look forward to learning about an AMA pilot with MSD in the Supply and Logistics track; get your tickets to join us there and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 9, 2024 | Technology |
In October 2024, CEPI announced that it is awarding funding of up to £3.7 million to support researchers at the University of Sheffield as they seek proof-of-concept for RNAbox. RNAbox is a specialised process designed to scale up the production of mRNA vaccines at regional vaccine sites. It is “easily adaptable and automated”, with the potential to improve global pandemic readiness by enabling increased equitable access to various mRNA vaccines, as and when needed. It also could help speed up responses to future emerging outbreaks.
Addressing mRNA challenges
mRNA vaccines be “more rapidly tailored” to specific diseases or variants, and the technology “holds promise” for different illnesses, including emerging infectious diseases. However, mRNA vaccines are “expensive to manufacture at a high product quality” and require complex cold-chain storage and transportation infrastructure. This makes them “extremely difficult to deliver to remote areas or low-resource settings”.
The RNAbox presents a potential solution to these challenges through its bespoke manufacturing process, designed to overcome the need to deliver the vaccine by facilitating local manufacture at small production sites. The process will run continuously to create between seven and ten times more mRNA at a time and enable more efficient use of raw materials. RNAbox uses digital-twin technology, in which a virtual replica of the vaccine manufacturing process is modelled on a computer in real-time with smart sensors collecting data on the physical product.
CEPI’s interest
CEPI states that the “fast, optimised vaccine production is critical to the 100 Days Mission”. The investment will explore applying the technology to vaccine development for CEPI priority pathogens, including the viruses that cause deadly diseases like Ebola, Lassa fever, MERS, and Nipah. Ingrid Kromann, Acting Executive Director of Manufacturing and Supply Chain at CEPI suggested that the University’s “versatile” technology “builds on the ‘vaccine revolution’ experienced during the COVID-19 pandemic”.
“It aims to overcome a number of scientific hurdles which resulted in poorer countries facing devastating vaccine inequity by helping to make high-quality, low-cost vaccines quickly and easily close to the source of an outbreak.”
Dr Zoltán Kis, School of Chemical, Materials, and Biological Engineering at the University of Sheffield, reflected on the “importance of being prepared” with the “necessary tools”.
“We need to tackle outbreaks equitably around the world, as diseases can spread across country borders.”
The RNAbox will “accelerate the development of new vaccines” and “mass-manufacturing against a wide range of diseases”.
“This transformative technology can also be used to develop much-needed vaccines against a range of unmet needs during non-epidemic/pandemic times. In case of a new epidemic/pandemic, the RNAbox can be quickly adapted to produce vaccines to tackle outbreaks. This will enable vaccine development and manufacturing capacity locally in countries around the world to serve local needs.”
The researchers will work with vaccine manufacturers in low- and middle-income countries to ensure the technology is fit-for-purpose in lower-resource settings.
At the Congress in Barcelona this month we will hear from experts who are revolutionising mRNA vaccine production to ensure products are accessible. Join us there to learn more, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 7, 2024 | Global Health |
Gavi shared the 2023 Annual Progress Report in October 2024, highlighting that more than 1.3 million future deaths were averted in 2023 through Gavi-supported vaccination programmes. The report details progress on strategic goals and reveals that the number of children protected with routine childhood vaccines since 2000 has exceeded 1.1 billion. These milestones also have economic benefits for Gavi-supported countries; the report suggests that this totals US$ 52 billion since 2021.
Chair of the Gavi Board, José Manuel Barroso, emphasised the importance of vaccinating children and vulnerable populations.
“We not only enable millions of people to lead healthier, more fulfilled lives [but we also] contribute to families’ prosperity, to strong and more stable communities, and to economic development that is already translating into countries’ paying more towards their immunisation programmes than ever before.”
Dr Sania Nishtar, Gavi’s CEO, commented that many Gavi countries are “on the front line of climate change, with many vulnerable to economic instability and geopolitical tension”.
“For them to be able to immunise more children, not to mention expand important programmes such as HPV, deserves recognition. Fully funding Gavi for its next five-year period will be crucial in expanding these hard-won gains and helping countries further along the pathway to fully sustaining their own immunisation programmes.”
Indicators and goals
Gavi partners and countries are “on track” to achieve most of the six mission indicators of the 2021-2025 strategic period:
- Under-five mortality rate
- Future deaths averted with Gavi support
- Future DALYs averted
- Reduction in number of zero-dose children
- Unique children immunised through routine immunisation with Gavi support
- Economic benefits generated through Gavi-supported immunisations
The mission is supported by four strategic goals
- Introduce and scale up vaccines
- Strengthen health systems to increase equity in immunisation
- Improve sustainability of immunisation programmes
- Ensure healthy markets for vaccines and related products
Vaccines
National Immunisation Coverage estimates in July 2024 confirmed that Gavi is on track in reaching children with new vaccines but must increase efforts to reach zero-dose and under-immunised children. At the end of 2023, Gavi had helped countries reach more than 1.1 billion children with routine immunisations since 2000. This means that the Investment Opportunity 2021-2025 commitment was achieved two years early. Gavi-supported countries completed a total of 13 routine introductions, taking the total introductions from 2021-2023 to 42.
Coverage of the third dose of diphtheria, tetanus, and pertussis-containing vaccine (DTP3) in 57 lower-income Gavi-supported countries remained “stable” at 80%. Apart from the pentavalent vaccine, Gavi-supported vaccines had higher coverage in 2023 than before the pandemic in 2019. After the opening of the support window for the second dose of inactivated polio vaccine (IPV2) in 2021, overall coverage in Gavi-supported countries increased rapidly to 27% by the end of 2023. The revitalisation of the HPV vaccine programme had “significant” effects: countries fully immunised more than 14 million girls with Gavi support in 2023.
Gavi’s vaccine portfolio has “grown significantly” over time; Gavi now supports vaccines against 20 infectious diseases through 53 product presentations.
Strategy indicators
Breadth of protection: In 2023 the 57 Gavi-supported countries (Gavi57) increased breadth of protection by 3 percentage points to 56%, against an implied target of 60% by 2025.
Coverage: Across the four vaccines included in the Sustainable Development Goal (SDG) indicator 3.b.1, the third dose of pneumococcal conjugate vaccine (PCV3) and the last dose in the schedule of human papillomavirus vaccine (HPVC) were trending higher in 2023 than originally projected. However, coverage of the second dose of measles-containing vaccine (MCV2) was “slightly behind but improving” and coverage of the third dose of DTP3 is “off track”.
Rate of scale up of new vaccines: Coverage of three vaccines (yellow fever: 97%, PCV: 93%, and rotaC: 93%) exceeded the benchmark. RotaC recovered from 2022 supply disruptions. Coverage of MCV2 remained under the 90% relative coverage target.
Introductions: 13 new routine introductions took place in 2023 against a milestone of 21. The cumulative total for introductions in 2021-2023 is 42, just “moderately delayed” against the target of 82 by 2025.
Country prioritisation: Gavi Secretariat considered if funding applications presented the three criteria (disease burden, effectiveness of vaccination, accounting for budget to meet requirements for vaccine procurement and sustain immunisation levels after transition from Gavi support). 93% of applications considered disease burden and increase in budget needed; 76% considered effectiveness of vaccination. 41 applications were reviewed from 2021 to 2023, increasing as countries submitted malaria vaccine applications.
Measles: 75% of children aged under five who were previously unvaccinated against measles received an MCV dose among countries conducting a Gavi-supported preventing MCV campaign.
Timely detection and response: Detection and response challenges, including “suboptimal surveillance” and lack of “robust” preparedness plans and locally available resources “persisted” in 2023. However, 5 out of 28 Gavi-supported outbreak responses with timeliness data met the disease-specific timeliness threshold in 2023. Measles-containing and yellow fever vaccines achieved higher rates of timely response than cholera, Ebola, and meningitis vaccines.
The future
Commenting on the progress presented in the report, UNICEF Executive Director Catherine Russell affirmed that “no child should die from vaccine-preventable diseases”.
“Through Gavi, the Vaccine Alliance we continue to bridge the gap between life-saving vaccines and the children who need them.”
To achieve the goals of the next strategic period, 2026-2030, Gavi needs to meet the funding target of US$9 billion. This will enable the organisation to expand protection against more diseases, ensure that the most vulnerable populations are “not left behind”, and protect the world against disease outbreaks. WHO Director-General Dr Tedros Adhanom Ghebreyesus stated that “vaccines are among the most powerful inventions in history”.
“With continued and increased investment in Gavi, we can harness their power, saving millions of lives in the coming decades.”
How do you think Gavi can continue to make immunisation progress into its next strategic period? What are the key challenges it faces? For more on the biggest vaccine challenges and opportunities to overcome them, join us at the Congress in Barcelona this month or subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 7, 2024 | Technology |
In October 2024, WHO announced that a fourth WHO-prequalified human papillomavirus (HPV) vaccine product, Cecolin, has been confirmed for use in a single-dose schedule. This update is an “important milestone” that will contribute to “improving sustainable supply of HPV vaccines”, ensuring that more people get access to the vaccines that prevent cervical cancer. HPV vaccine programmes have been “hampered” by supply shortages since 2018, and recent production challenges have led to further shortfalls, which will affect girls in need of HPV vaccines Africa and Asia.
Eliminating cervical cancer by tackling HPV
More than 95% of the 660,000 annual cervical cases are caused by HPV. Every two minutes, a woman dies from the disease, and 90% of these deaths happen in low- and middle-income countries. 19 out of the 20 “hardest hit” countries are in Africa. However, vaccination is an effective way of addressing this health need. Dr Tedros Adhanom Ghebreyesus, WHO Director-General, states that “we have the ability to eliminate cervical cancer, along with its painful inequities”.
“By adding another option for a one-dose HPV vaccination schedule, we have taken another step closer in consigning cervical cancer to history.”
Dr Kate O’Brien, Director of the Department of Immunisation, Vaccines, and Biologicals at WHO, reflected that achieving a 90% coverage in girls by the age of 15 is the target of the first pillar of WHO’s global strategy for cervical cancer elimination.
“Given the continuing supply challenges, this addition of single dose vaccine product means countries will have greater choice of vaccines to reach more girls.”
Cecolin
Cecolin is a bivalent HPV vaccine delivered intramuscularly as a single dose. It is manufactured by Xiamen Innovax Biotech and should be stored between 2°C and 8°C. It is designed to protect against HPV types 16 and 18, which are commonly associated with the development of cancer. When Cecolin received prequalification, PATH stated that it had provided “technical assistance” for the process to facilitate greater accessibility. PATH’s China country representative Yuan Yuan commented that the vaccine would put the world “on its way to more equitable HPV vaccination”.
Single-dose coverage
Several products that were initially prequalified for use in a 2-dose schedule can now be used in a single-dose schedule. Cecolin can be recommended for “off-label” use after data support the modified schedule until the manufacturer adds the modified use to the label. Data from July 2024 show an increase in one dose HPV vaccine coverage among girls aged 9-14 years, from 20% in 2022 to 27% in 2023. In 2023, 37 countries were implementing a single-dose schedule; this increased to 57 by September 2024. WHO suggests that the adoption of a single-dose schedule has resulted in “at least” 6 million additional girls being reached with HPV vaccines in 2023.
For insights into the role of vaccination in elimination strategies, why not join us at the Congress in Barcelona this month? Don’t forget to subscribe to our weekly newsletters here for more vaccine news.

by Charlotte Kilpatrick | Oct 7, 2024 | Global Health |
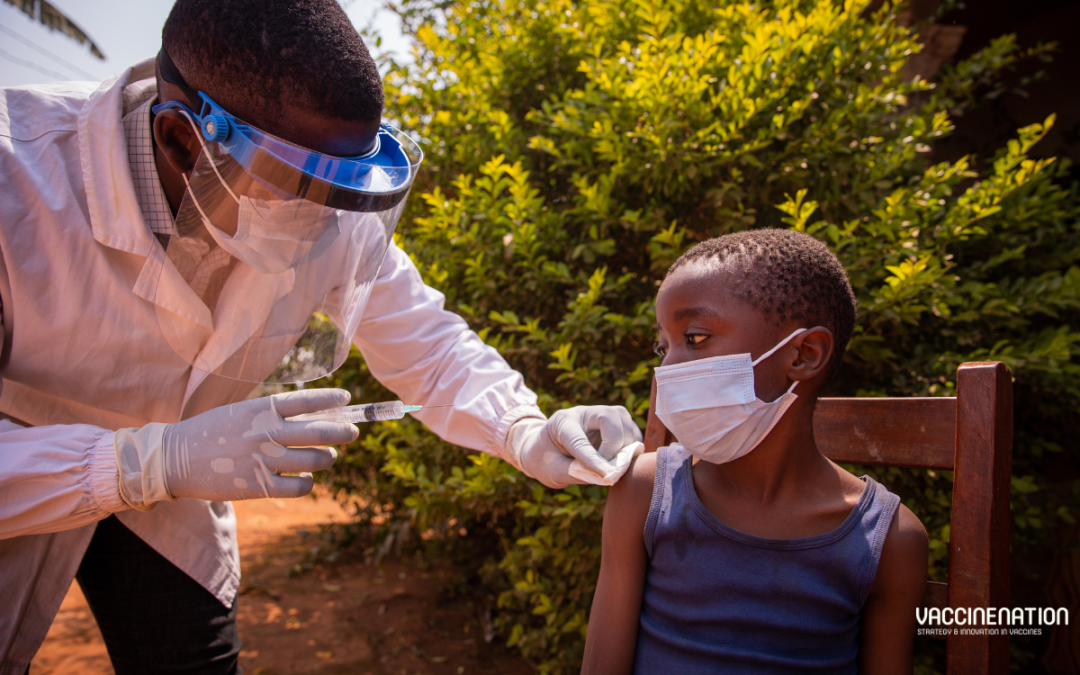
WHO Africa reported in October 2024 that the Democratic Republic of the Congo (DRC) has started a vaccination campaign as part of outbreak control efforts against mpox. The vaccination drive has launched in the eastern North Kivu province and will prioritise health workers and frontline responders, contacts of confirmed cases, contacts of those contacts, and other at-risk groups. It will later be implemented in 11 of the most affected health zones in Equateur, North Kivu, Sankiri, South Kivu, Sud-Ubangi, and Tshopo provinces.
Addressing the emergency
The Democratic Republic of the Congo (DRC) has reported more than 30,000 suspected and laboratory-confirmed cases and 990 deaths since the start of 2024. These numbers account for 90% of the cases reported from 15 countries in the African region this year. WHO recommends that vaccination should form part of a “comprehensive response” involving enhanced surveillance, community engagement, and case management. It is working with partners and the national authorities to “scale up and reinforce all the key control measures”.
In preparation for the mpox vaccination campaign, WHO has supported national health authorities in training health workers, enhancing vaccine delivery systems and infrastructure, and community engagement. There are also efforts to “reinforce measures to identify and address” vaccine misinformation and disinformation, responding with increased access to accurate information.
Delivering doses to affected areas
Noting that mpox vaccines are “currently in short supply, especially in Africa”, WHO is encouraging global collaboration to get doses to the people who need them most. In September, WHO prequalified MVA-BN for mpox, which is “expected to facilitate timely and increased access”. It is also working with partners like Gavi and UNICEF to establish a distribution mechanism for donated doses and direct procurements. DRC has received 265,000 doses of MVA-BN, donated by the European Commission’s Health Emergency Preparedness and Response Authority, Gavi, and the United States Government.
WHO Regional Director for Africa, Dr Matshidiso Moeti, expressed gratitude to these partners for their donations.
“As we rally efforts to stop the mpox outbreak, the rollout of the vaccine marks an important step in limiting the spread of the virus and ensuring the safety of families and communities.”
Dr Moeti commented that WHO is “working closely with the national authorities to effectively deliver the vaccines to those who need them most”. Africa CDC also recognised the collaborative effort, which “underscores the collective global commitment” to controlling the outbreak in Africa. H.E. Dr Jean Kaseya, Africa CDC Director General, commended the DRC’s “swift action” in launching the campaign, which “showcases the strength of its public health leadership”.
“By prioritising vulnerable populations, including frontline health workers and those most at risk, the country is taking critical steps to contain the outbreak. Africa CDC remains committed to working closely with the DRC to ensure vaccines reach those who need them the most, while also working to strengthen health systems to prevent future outbreaks. Our top priority is to secure safe and effective vaccines for children in the next phase of vaccination.”
Mpox vaccination will be a key area of high-level discussions at the Congress in Washington next April, including on a keynote panel that will consider the “role of vaccines in a changing world”. Get your tickets to join us for these conversations, and don’t forget to subscribe to weekly vaccine updates here.

by Charlotte Kilpatrick | Sep 27, 2024 | Global Health |
WHO and TikTok announced a year-long collaboration to provide “reliable, science-based health information” in September 2024. The partnership seeks to address the challenges of misinformation and disinformation on digital channels by “promoting evidence-based content and encouraging positive health dialogues”. TikTok is a social platform where users create and share short-form videos.
The Fides network, a “network of healthcare influencers” who seek to share “good health content” and tackle misinformation, was launched in 2020. It has over 800 creators with a reach of 150 million people on various platforms. Network creators across the globe will be joining TikTok to create and promote evidence-based content. TikTok is also making a $3 million donation to support WHO’s work on “destigmatising mental health conditions and creating an informed, empathetic, and supportive online community”.
Social channels as a source
WHO recognises that social media platforms are important sources of information that can influence health-related behaviours and decisions. It states that one in four young adults seeks news content on social media platforms such as TikTok. However, these digital channels are increasingly allowing the distribution of misinformation and “malinformation”. Thus, the collaboration will “expand efforts” on several health topics, making science-based information “relatable and digestible”, and offering support for influencers through TikTok’s creator training programmes.
WHO’s Chief Scientist Dr Jeremy Farrar hopes that the collaboration will prove to be an “inflection point in how platforms can be more socially responsible”.
“The intersection of health and technology presents an opportunity to reach people of all ages, where they are, when they want to access. By working with TikTok and others, we are helping people access credible information and engage in scientific discourse that collectively helps shape a healthier future for all.”
Dr Alain Labrique, WHO’s Director of Digital Health and Innovation, reflected that “creators who understand their audience’s needs have a unique opportunity to bridge the gap between science and everyday life”.
“This is where WHO can step in to support influencers in delivering evidence-based information, ensuring that health conversations on platforms like TikTok are both impactful and informed.”
TikTok’s Global Head of Trust and Safety, Outreach and Partnerships, Valiant Richey, commented on the importance of TikTok’s commitment to providing “reliable information”.
“We are delighted to be partnering with the World Health Organisation’s Fides network of healthcare content creators to further strengthen this commitment by bringing engaging and authoritative mental well-being content to our community.”
Creators leading the field
Dr Timothy Tiutan has created a community of almost 2 million followers on social media and hopes that the initiative will enable creators to “empower communities to live healthier lives”.
“The network tackles global health challenges in an era where access to health information has dramatically evolved. WHO Fides is a driving force in shaping a healthier, more informed global community for the future.”
Avisha NessAiver specialises in translating research into accessible language and has worked with Fides and the UN as part of “Team Halo”. His content has reached over 100 million views on various platforms.
“The Fides network is the catalyst transforming isolated scientists and health experts into a powerful collective force, armed with shared knowledge and strategies to effectively combat the spread of health misinformation.”
Do you think this initiative will be an effective way of engaging social media users in reliable information? Or will the partnership ruffle feathers online and in the lab?
To discuss the importance of effective communication and translating the latest research into accessible content with your colleagues at the Congress in Barcelona next month, get your tickets here, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 26, 2024 | Technology |
UNICEF announced in September 2024 that it is working with Australia and Samoa to improve access to vaccinations in Samoa. To this end, an innovative new vaccine delivery vehicle was delivered, alongside waste management supplies such as bins and masks. These supplies are intended for the benefit of everyone living in Samoa, including healthcare providers in 13 healthcare facilities.
A vehicular ‘boost’
UNICEF describes the arrival of the Pacific nation’s first vaccine delivery vehicle as a “boost” to the health system, with positive implications for cost effectiveness and wastage of resources for healthcare workers. Until this point, a smaller vehicle has been used to transport vaccines from the airport to the National Vaccine Centre, before distribution to health care facilities. This required more trips to ensure the potency and efficiency of the vaccines was maintained.
Although Samoa has been making “steady progress” in reaching eligible children with vaccines in the national immunisation schedule and has received support from UNICEF on cold chain and capacity building, the transportation and distribution of vaccines has been identified as a “gap”. The new vehicle, procured with support from the Australian Government and UNICEF, will enable faster transportation and can protect vaccines in unfavourable weather conditions.
Strengthening the health system
Samoa’s Hon. Minister of Health, Valasi Luapitofanua To’ogamaga Tafito Selesele, expressed gratitude to the Australian Government for “timely support” and UNICEF for “able technical guidance and support”, which will help to strengthen the health system to “get equipped to provide quality services in a cost-effective way”. Australia’s High Commissioner to Samoa, H.E. Will Robinson, stated that the vehicle, although prioritised for vaccines, can be used to “accommodate the pharmaceutical supplies for distribution” to health facilities.
“Australia is proud to be a long-term partner for Samoa in achieving its vision for a healthier community and delivering better health for all.”
UNICEF Pacific’s Chief of Samoa Field Office, Khin Moe Aye, recognised that “most” of the health issues facing children in Samoa and the Pacific are preventable.
“However, preventing them requires an effective primary healthcare system well-supported by sound resources. UNICEF is pleased to partner with the Australian Government and Ministry of Health in Samoa towards the strengthening of healthcare systems. This will enable children and their families to access quality services, while also ensuring that healthcare workers benefit from better resources and services.”
For the latest insights into strengthening healthcare systems and ensuring effective vaccine delivery strategies across the world, join us at the Congress in Barcelona next month. Don’t forget to subscribe to our weekly newsletters for more!

by Charlotte Kilpatrick | Sep 26, 2024 | Global Health |
In September 2024 Africa CDC and IAVI announced the signing of a Memorandum of Understanding (MoU) to enhance the continent’s capacity to fight disease, pandemic readiness, and supply resilience. This will involve expanding capabilities for locally driven research, development, manufacturing, and supply of priority vaccines and antibodies as well as strengthening Africa CDC-led initiatives. The partnership will combine IAVI’s “expertise in vaccine and antibody development and access” and the “extensive network and Africa CDC”.
Initiatives under the MoU
The MoU is intended to tackle pressing public health challenges and promote long-term health security. Some of the key initiatives under the MoU include:
- Supporting the development of vaccines and antibodies for regional health priorities (like Lassa fever and HIV)
- Fostering a sustainable supply and demand ecosystem for priority products in the region (including monoclonal antibodies)
- Strengthening African research and development capacity
- Exploring regional stockpile strategies for licensed and investigational products to ensure rapid responses during health crises
The MoU exemplifies the “action-oriented partnerships” that Africa CDC’s New Public Health Order demands as the organisation drives its vision for “redefining global health architecture” and ensuring that Africa and the world are better prepared for future health threats. IAVI recognises the support of funders and partners, including Wellcome, CEPI, the European and Developing Countries Clinical Trials Partnership (EDCTP), the United States Agency for International Development (USAID) and the United States President’s Emergency Plan for AIDS Relief (PEPFAR) through the Accelerate the Development of Vaccines and New Technologies to Combat the AIDS Epidemic (ADVANCE) programme.
Dr Mark Feinberg, IAVI President and CEO, described the cooperation as a “key step” in IAVI’s mission to “improve global access to biomedical innovations and safeguard public health”.
“It goes beyond R&D; it’s about creating a vibrant health innovation ecosystem that meets current and future needs across Africa.”
We look forward to welcoming senior representatives of IAVI back to the Congress in Barcelona next month to learn more about the various efforts and initiatives they are enabling in pursuit of global health goals. Get your tickets to join us there and don’t forget to subscribe to our weekly newsletters here.

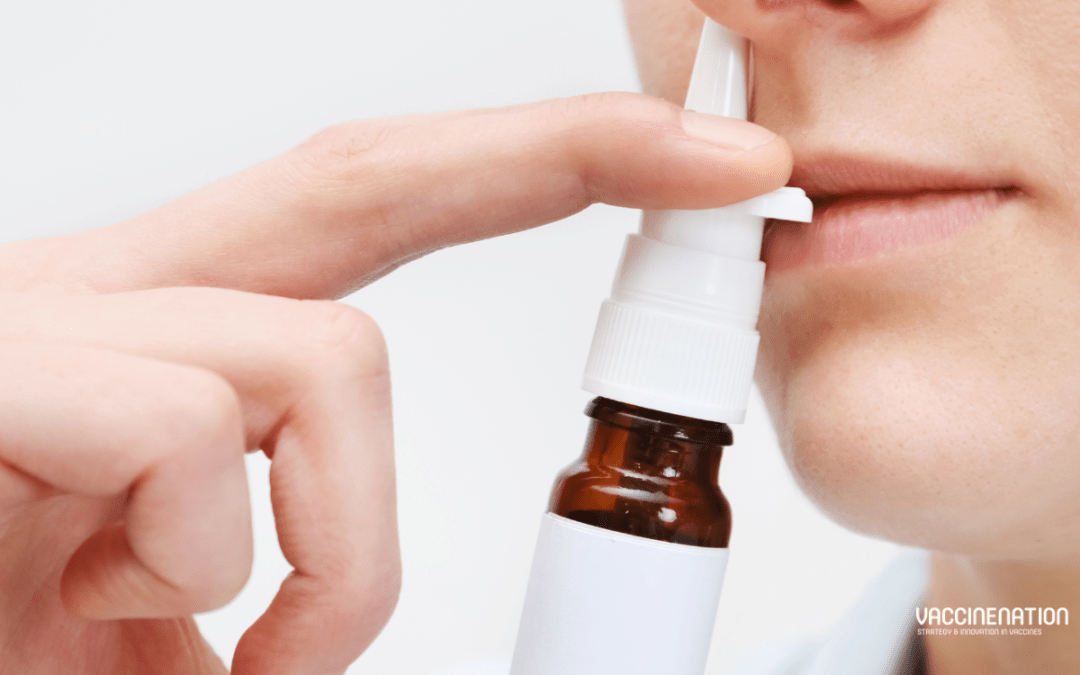
by Charlotte Kilpatrick | Sep 23, 2024 | Technology |
In September 2024 the United States FDA announced the approval of FluMist for self- or caregiver-administration. It is approved for active immunisation for the prevention of influenza disease caused by subtypes A and B in individuals aged 2 to 49. FluMist is administered as a nasal spray and was initially approved for use in 2003, with an expansion to include younger children in 2007. The latest approval was based on a submission that included results from a usability study, which demonstrated that individuals over the age of 18 could self-administer or administer FluMist to eligible recipients. It is the first vaccine to prevent influenza that does not require administration by a health care provider.
FluMist
The live attenuated influenza vaccine (LAIV) is administered as a nasal spray. The vaccine is recommended by the Advisory Committee on Immunisation Practices (ACIP) and American Academy of Paediatrics (AAP). A prescription is required to receive FluMist, but the vaccine can either be administered by a health care provider in a health care setting or by the vaccine recipient or caregiver over 18. Since its initial approval in the United States in 2003, almost 200 million doses have been distributed globally.
In FDA-required studies, AstraZeneca evaluated if individuals from 18 to 49 years of age could “appropriately” administer FluMist when given instructions. These studies showed that 100% of intended users administered a full dose, and that the efficacy, immunogenicity, and adverse events with self-administration were “similar to” those seen with administration in a health care setting.
The vaccine manufacturer will make the vaccine available through a third-party online pharmacy for people who are interested in self- or caregiver-administration. To order FluMist, these people will complete a screening and eligibility assessment before receiving a prescription and delivery of the vaccine. Individuals aged 2 to 17 should not self-administer.
Broader accessibility
Director of the FDA’s Centre for Biologics Evaluation and Research Dr Peter Marks commented that the approval provides a “new option” for safe and effective seasonal influenza vaccination, “potentially with greater convenience, flexibility, and accessibility for individuals and families”.
“Getting vaccinated each year is the best way to prevent influenza, which causes illness in a substantial proportion of the U.S. population every year and may result in serious complications, including hospitalisation and death. This approval adds another option for vaccination against influenza disease and demonstrates the FDA’s commitment to advancing public health.”
Dr Ravi Jhaveri, Division Head, Infectious Disease, Virginia H. Rogers Professor in Infectious Diseases, Professor of Paediatrics, Northwestern University School of Medicine, also emphasised the importance of increasing accessing to vaccinations to address the “significant burden” of influenza.
“For the first time, families and caregivers will be able to protect themselves against influenza with a needle-free, self-administered vaccine, from the convenience of their own home.”
Iskra Reic, Executive Vice President, Vaccines and Immune Therapies, AstraZeneca, stated that the approval of FluMist for self-administration is an “important step forward in making vaccines more accessible to fight the high annual burden of influenza”.
“For more than 20 years, FluMist has been the only nasal spray flu vaccine licensed in the US and now it is also the only vaccine to help individuals, families, and communities access an influenza vaccine conveniently through self- and caregiver-administration outside of traditional healthcare settings.”
To learn more about the various applications of nasal spray vaccines at the Congress in Barcelona next month, get your tickets to join us here. Don’t forget to subscribe to our weekly newsletter for more vaccine insights.

by Charlotte Kilpatrick | Sep 20, 2024 | Global Health |
In September 2024, Africa CDC offered congratulations to the Governments of Japan and the Democratic Republic of the Congo (DRC) for their agreement on the donation of Lc16 mpox vaccines and specialised inoculation needles. At a signing ceremony in Kinshasa, the two governments marked a “significant milestone” in their cooperation. These vaccines are a “critical boost” to DRC’s mpox response; Lc16 is the only mpox vaccine currently approved for children.
Lc16
The vaccine is derived from the Lister strain of vaccinia and contrasts to the replication-deficient vaccines like Modified Vaccine Ankara (MVA) by retaining most of vaccinia genome and being able to replicate at the site of inoculation. It is administered as a single dose through the scarification method, which involves scratching the skin before applying the vaccine solution. Studies in children suggested that the vaccine was safe and well tolerated, leading to licensure. However, the duration of immunity remains unclear.
Another tool for the fight
Africa CDC states that this donation comes “at a pivotal time” in DRC’s fight against the regional mpox outbreak. Since the declaration of a PHECS in August 2024, a “more coordinated international response” has evolved. Central to this response is vaccination, which is complicated by Africa’s “limited access to critical countermeasures”.
Director General of Africa CDC, H.E. Dr Jean Kaseya, is “deeply appreciative” of the ‘” of the “generous donation” of mpox vaccines and specialised vaccine needles to the DRC.
“This timely assistance will significantly bolster our ongoing efforts to contain the outbreak, and I am confident that this partnership will help mitigate the public health threat posed by mpox, not only in the DRC but across the continent.”
Japan’s Senior Deputy Minister for Foreign Affairs, Takeshi Akahori notes the increasing number of cases in the DRC and other countries, commenting that Japan is “monitoring the situation closely” with WHO.
“I hope that these vaccines and needles will contribute meaningfully to the fight against mpox.”
Join us at the Congress in Barcelona next month to engage with public health experts on current health threats and how vaccines can contribute to our response, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 19, 2024 | Infection |
The Times of India reported in September 2024 that cases of chikungunya in children were symptomatic of a “shift” in epidemiology. Case numbers have increased and are more frequently requiring hospitalisation, particularly among children. While these changes are cause for concern for some infectious disease experts, they are careful to emphasise the need for thorough research. Indeed, the increase in cases may be attributed to improved surveillance efforts.
Cases in children
The latest report suggests that in Maharashtra, a populous state in the west of India, cases have exceeded 2,600 since the start of the year. This is a 50% increase on the previous year, and the disease is “increasingly” affecting children, some of whom require time in intensive care. Dr Sachin Shah, neonatologist at Surya Hospital in Pune, is concerned by the number of children suffering:
“In my OPD for older children, it is the most common complaint this week.”
Five newborns were diagnosed with chikungunya in Pune, two of whom acquired it from their mothers. Dr Shah described high-grade fever, elevated liver function, and low appetite among the symptoms. One neonate developed a “life-threatening disorder” characterised by hyper-inflammation. Infectious disease specialist at H N Reliance Hospital in Girgaum notes the change in severity among patients who are admitted to hospital.
“Chikungunya has acquired a notorious edge this year.”
This change is also observed by Dr Anita Mathew, infectious disease specialist at Fortis Hospital, Mulund, who has had patients require ICU attention.
“The extreme symptoms are new to Mumbai, but are not unknown in medical literature.”
Is this new?
While some experts are questioning this “shift” in disease, others are wary of sounding the alarm. Dr Radhakrishna Pawar of the state health department acknowledged the change in clinical appearance but urged further investigation.
“We need to study it in detail before making such claims.”
Furthermore, while cases have increased, Dr Pawar noted that they do not reach the numbers required to qualify as an outbreak or epidemic.
Vaccine access
Although there is a licensed chikungunya vaccine to address the “significant unmet medical need” presented by chikungunya, Valenva’s IXCHIQ is only currently approved in the United States, Europe, and Canada. However, Valneva is “focussed on expanding the vaccine’s label and access”, working with CEPI to support access in LMICs, post-marketing trials, and potential label extensions.
For the latest on infectious disease management with vaccine innovation, join us at the Congress in Barcelona next month, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 18, 2024 | Global Health |
In September 2024, Gavi and Bavarian Nordic announced an advance purchase agreement (APA) to secure 500,000 doses of MVA-BN mpox vaccine to be supplied to countries in Africa that are affected by the mpox outbreak. The doses are funded by Gavi’s First Response Fund and are for delivery in 2024. Bavarian Nordic will be ready to supply the vaccine doses after a supply agreement has been signed with UNICEF, which will deliver the doses.
The First Response Fund
Gavi’s First Response Fund was established in June 2024 to “make cash rapidly available” for the purchase of vaccines in health emergencies. It is available to Gavi-eligible countries in the early days of a pandemic or major health emergency. The Fund pre-positions up to US$500 million of surge financing for vaccine procurement ‘at risk’, which means funds are used to secure doses and “Gavi’s place in the queue” while manufacturers complete the final steps of regulatory approval and manufacturing scale-up.
Dr Sania Nishtar, Gavi’s CEO, commented that the First Response Fund was designed in collaboration with donors and partners “specifically to provide rapid early funding for emergencies such as mpox”.
“Using it today to fund the first direct transaction for vaccines in support of equitable access and the global response, just over a month since mpox was declared a public health emergency, takes us a long way towards our goal of protecting those most at risk.”
Dr Nishtar thanked donors for enabling Gavi to “act rapidly” by committing funds before an emergency occurred.
“We are committed to working with affected governments and our partners to turn these vaccines into vaccinations as quickly and effectively as possible and, over time, to build a global vaccine stockpile if sufficient funding is secured for Gavi’s work through 2030.”
Paul Chaplin, President and CEO of Bavarian Nordic, is pleased to sign the agreement and “strengthen our commitment to support Gavi and other global health partners” who demonstrate “strong leadership”.
“The doses secured through this agreement will significantly increase the availability of mpox vaccines for African countries, and we are pleased that Gavi has selected our MVA-BN vaccine, which has proven highly effective during the global mpox outbreak in 2022.”
For the latest on vaccine collaborations to ensure equitable access, get your tickets to join us at the Congress in Barcelona next month, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 16, 2024 | Global Health |
In September 2024 WHO announced the establishment of an access and allocation mechanism for mpox medical countermeasures, including vaccines, treatments, and diagnostic tests. The Access and Allocation Mechanism (AAM) is intended to increase access to these essential tools for people at highest risk, ensuring that limited supplies are used “effectively and equitably”. This announcement comes after WHO declared the mpox outbreak a PHEIC in August 2024 and addresses one of the key International Health Regulations Emergency Committee’s recommendations: “equitable access to safe, effective, and quality-assured countermeasures”.
AAM
The AAM is part of the interim Medical Countermeasures Network (i-MCM-Net). Developed in response to “global vulnerabilities” exposed by the COVID-19 pandemic, i-MCM-Net enhances collaboration through a “Network of Networks” approach. It seeks to provide timely and equitable access to quality, safe, effective, and affordable medical countermeasures in response to public health emergencies through existing networks and global collaboration. The network was endorsed by WHO Member States as an interim mechanism while negotiations on a pandemic agreement continue.
The mpox AAM includes members of the i-MCM-Net as well as WHO: Africa CDC, CEPI, the EU Health Emergency Preparedness and Response Authority (HERA), FIND, Gavi, the PAHO Revolving Fund, UNICEF, Unitaid, and others. It will work to allocate the “currently scarce supplies” to those at highest risk of infection.
It will operate according to three guiding principles:
- Preventing illness and death – prioritise vaccination and other tools to interrupt transmission for those at greatest risk to prevent illness and death.
- Mitigating inequity – ensure equitable access to medical countermeasures for all people at risk, irrespective of socio-economic or demographic background.
- Ensuring transparency and flexibility – establish and maintain clear and open communication about allocation decisions and be ready to adapt strategies as new data emerge or situations change.
More than 3.6 million vaccine doses have been pledged for the mpox response, including 620,000 doses of MVA-BN pledged to affected countries by the European Commission, Austria, Belgium, Croatia, Cyprus, France, Germany, Luxembourg, Malta, Poland, Spain, and the United States of America, as well as Bavarian Nordic. Japan has pledged 3 million doses of the LC16 vaccine. This is the largest pledge so far.
International coordination
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, recognised the need for “powerful tools” like vaccines, therapeutics, and diagnostics, to bring the mpox outbreak “under control”.
“The COVID-19 pandemic illustrated the need for international coordination to promote equitable access to these tools so they can be used most effectively where they are most needed. We urge countries with supplies of vaccines and other products to come forward with donations, to prevent infections, stop transmission, and save lives.”
Dr Mike Ryan, Executive Director of WHO’s Health Emergencies Programme, emphasised that WHO and its partners are working with the government of the Democratic Republic of the Congo and other affected countries to “implement an integrated approach to case detection, contact tracing, targeted vaccination, clinical and home care, infection prevention and control, community engagement and mobilisation, and specialised logistical support”.
“The AAM will provide a reliable pipeline of vaccines and other tools in order to ensure the success on the ground in interrupting transmission and reducing suffering.”
Join us at the Congress in Barcelona next month to share your insights on the best ways to ensure equitable access to essential medical countermeasures, and don’t forget to subscribe to our weekly newsletters for the latest vaccine news.