by Charlotte Kilpatrick | Oct 23, 2024 | Technology |
The National Institute for Health and Care Research (NIHR) announced in October 2024 that the first Phase III randomised trial of an investigational mRNA norovirus vaccine is opening across the UK. The trial seeks to evaluate the efficacy and safety of mRNA-1403 and is sponsored by Moderna. It will be conducted across 39 sites in the country, including 27 NHS primary and secondary care sites. Investigators hope to recruit 2,500 participants between late October and early 2025.
Norovirus
Norovirus, or the “winter vomiting bug”, is a viral illness; it is the “most common cause” of acute gastroenteritis globally. Symptoms include acute onset diarrhoea and vomiting, but there is evidence to suggest that norovirus infection is associated with intestinal inflammation and malnutrition and could cause long-term morbidity. WHO estimates that 685 million cases of norovirus are seen each year, including 200 million cases in children under 5.
“The burden of norovirus is significant; norovirus causes an estimated 200,000 deaths per year, including 50,000 child deaths, primarily impacting low-income countries. Norovirus has been estimated to cost $60 billion globally as a result of healthcare costs and economic losses.”
There are no licensed norovirus vaccines in use.
The trial
Participants in the trial will be randomly assigned to two groups. One group will receive the investigational vaccine, and the other groups will receive a placebo. While anyone who is aged 18 or older and meets the inclusion criteria is eligible for the trial, researchers are seeking participants aged 60 and older. This age group is “generally more likely to be severely affected by norovirus”. Thus, a successful vaccine would be of greatest benefit to them.
The trial is part of the 10-year partnership between Moderna and the UK Health Security Agency (UKHSA) on behalf of the UK Government. It is supported by the UK Vaccine Innovation Pathway (VIP) and the NIHR, which are working together to accelerate vaccine trials in the UK. The NIHR Research Delivery Network (RDN) is working with the devolved administrations to accelerate the roll-out of “key” vaccine studies. These efforts include the NIHR’s flagship recruitment tool: Be Part of Research.
Be Part of Research is an online service that connects people to health and care research across the UK. Anyone over the age of 18 can register for an account, indicating the areas of research that they are interested in. They are then sent information about suitable studies.
Advancing healthcare
The UK Chief Investigator, Dr Patrick, is a GP in Dorset and Research Director at the NIHR’s South West Regional Research Delivery Network. Dr Moore commented that the UK is “really pleased to be able to play an important role” in finding a vaccine for this “highly contagious disease”.
“Outbreaks of norovirus have huge consequences, both on our health systems and our economy. This innovative trial is crucial in helping us advance healthcare.”
NIHR Chief Executive and Chief Scientific Advisor to the Department of Health and Social Care, Professor Lucy Chappell, hopes that the vaccine could “make a difference to lives of many – especially our most vulnerable citizens”.
“Leveraging the UK’s expertise in vaccine development, the DHSC through the NIHR and Moderna are delivering this large-scale trial at pace, so that people across the UK and the world can benefit sooner.”
Health and Social Care Secretary Wes Streeting described the trial as a “huge vote of confidence in the UK’s life sciences sector”. It could also allow a “shift” away from sickness towards prevention, which would reduce pressure on the NHS and keep people healthier in the colder months. Moderna’s Chief Development Officer Dr Melanie Ivarsson is “delighted” to bring the trial to the UK.
“By advancing our investigational mRNA norovirus vaccine into a pivotal Phase III trial, we are one step closer to potentially providing a new tool to prevent infection from this highly contagious virus, which places a significant burden on health systems globally.”
Sarah Collins, Commercial Director at UKHSA, reflected that norovirus “isn’t just a nasty tummy bug – it can have serious consequences” including “disruption” to health services and education settings.
“It’s fantastic to see the first trial of its kind get underway as a result of UKHSA’s strategic 10-year partnership with Moderna, which aims to enhance investment in scientific research and vaccine development to enable a rapid response to future pandemics and other infectious diseases.”
For the latest on vaccine research, don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 17, 2024 | Global Health |
A study in The Lancet Global Health estimates the impact that the 100 Days Mission could have had on the COVID-19 pandemic. The authors find that the implementation of non-pharmaceutical interventions (NPIs) within the 100 Days Mission could have averted around 8.33 million deaths, corresponding to a monetary saving of US$14.35 trillion. Investment in manufacturing and health systems contribute an increase to 11.01 million deaths averted. The researchers comment on the value of the 100 Days Mission but emphasise the importance of “prioritising a more equitable global vaccine distribution”.
In search of greater vaccine benefits
Although COVID-19 vaccines are estimated to have prevented almost 20 million deaths, the authors demand a better understanding of the “further health and economic benefits that could have been achieved” through shorter development times and “improved global equity in pandemic preparedness”. CEPI’s 100 Days Mission was proposed in 2021, establishing the ambition of cutting vaccine development time for new pathogens to 100 days. This about a third of the time taken to deliver the first COVID-19 vaccine.
“The availability of COVID-19 vaccines within 100 days would have substantially changed the pandemic; however, these benefits would be finite without enabling equitable access to vaccine products through system equity.”
Various efforts to encourage global vaccine distribution were “hampered” by inequities, and it is recognised that manufacturing capacity should be “expanded but also diversified” to “promote self-sufficiency and regional resilience”. Furthermore, supply chains and infrastructure must be scaled to enable the delivery of vaccines that rely on cold-chain infrastructure.
The study
The searchers hoped to quantify the potential impact of the 100 Days Mission by retrospectively estimating the effect it would have had on the COVID-19 pandemic, thus offering evidence to support decision making around future investments in research and development capabilities. They also aimed to quantify the impact of “additional investments”.
The analyses use an extended version of a previously published compartmental susceptible-exposed-infectious-recovered transmission model of COVID-19 vaccination with an explicit healthcare pathway. The vaccination pathway was expanded to include booster vaccination alongside waning efficacy, capturing the “restoration of immunity” through booster doses. The new vaccination pathway was parameterised to match platform-specific vaccine efficacy data and the duration of protection.
The authors modelled the impact of the 100 Days Mission by simulating a counterfactual scenario where the global vaccination campaign began on 20th April 2020, 100 days after the publication of the full SARS-CoV-2 genome. This scenario assumes that vaccinations in each country took the same roll-out process, but 232 days earlier. Two additional scenarios reflected “increased investment” in research and delivery infrastructure.
The Manufacturing scenario removed supply constraints, enabling the rollout of vaccination on 20th April 2020 in every country, without stockouts. The infrastructure-equity scenario enhanced both national health systems and global distribution networks so that all countries achieved 40% vaccine coverage in the first year and 40% booster coverage in the second year.
To account for the relaxing of non-pharmaceutical interventions (NPIs), the authors simulated three scenarios for NPI relaxation speeds as vaccination coverage improved. The History scenario assumed no changes, whereas the Target and Economic scenarios assumed earlier relaxing; the Target scenario lifted all restrictions over two months after reaching more than 80% adult coverage in high-income countries or more than 80% coverage in those older than 60 in other countries. The Economic scenario lifted NPIs more gradually after reaching the over-60 target, prioritising the reopening of schools.
Study findings
The results suggest that the 100 Days Mission could have averted an additional 8.33 million deaths due to COVID-19 by the end of 2021 when combined with the History NPI lifting scenario. In this scenario, an estimated 26.72 million severe cases of COVID-19 requiring hospitalisation and 1/44 billion infections would have been averted. Most of these averted deaths, hospitalisations, and infections would have occurred in low- and middle-income countries (LMICs).
The estimated VSLs (value of a statistical life) that could have been saved by the 100 Days Mission through the History scenario is US$22.61 trillion globally. As VSLs are “significantly higher” in HICs, 57% of the global value of statistical life averted occurred in HICs, even though most deaths would have been averted in LMICs. To estimate the monetary values associated with lives saved the authors multiplied the number of lives saved by the country-specific monetary VSL and by the value of a statistical life-year (VSLY).
“Increased investment in both global manufacturing and health systems infrastructure further increases the number of deaths that could be averted and the associated health-economic savings.”
In the 100 Days Mission with both manufacturing and health systems investments, an estimated 11.01 million deaths could have been averted and a value of $31.29 trillion in statistical lives saved. However, the authors describe this scenario as “unlikely”. In all scenarios involving the relaxing of NPIs thanks to earlier availability of vaccines, additional lives would have been saved.
In the Target NPI lifting scenario, an estimated 5.76 million deaths (100 Days Mission alone) to 9.20 million deaths (100 Days Mission with both manufacturing and infrastructure investments) could have been averted. In these scenarios, 12,600 and 23,900 fewer days of NPIs would have been implemented globally: 70 days and 133 days on average per country. Under the Economic scenario there were “similar trade-offs between public health and economic gains”. The public health and health economic outcomes would be greater than under the Target scenario, but still lower than the History scenarios.
Substantial benefits
“Earlier access to COVID-19 vaccines could have had substantial benefits.”
Most of the estimated averted deaths would have been concentrated in LMICs, but this demands investments in vaccine research, supported by “improvements to manufacturing and health system infrastructures”. With these investments, the authors estimate that 11 million deaths could have been prevented globally.
Although NPIs were effective at reducing transmission they incurred “significant economic and societal costs”, including consequences for education. Therefore, a major benefit of earlier access to vaccination is the reduction in school closures; in the Economic scenario, prioritising school opening could have averted 1,120 weeks of full school closures and 2,490 of partial school closures. This represents an average of 6 weeks of fully open schools and 14 weeks of partly open schools per country.
“Reopening schools and relaxing NPIs safely will crucially require scaling up both vaccine delivery infrastructure and manufacturing. Without addressing both aspects, advancements in vaccine development speed might not translate into equitable benefits globally.”
The results emphasise the importance of investments in support of the 100 Days Mission in controlling a future potential pandemic, with benefits for both health and economy.
“The 100 Days Mission is ambitious, requiring global innovation through creating vaccine libraries, clinical trial networks, accelerated immune response marker identification, rapid vaccine manufacturing, and strengthened global disease surveillance.”
CEO of CEPI, Dr Richard Hatchett, hopes that this research will encourage global commitment to the 100 Days Mission.
“This work shows in the starkest terms why the world needs to be prepared to move faster and more equitably when novel pandemic disease threats emerge. Investing in preparedness now to make the 100 Days Mission possible for future incipient pandemics will save millions upon millions of lives and protect the global economy against catastrophic losses.”
Join us at the Congress in Barcelona this month to explore how lessons from the COVID-19 pandemic can inform better global preparedness for pandemic and epidemic pressures, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 14, 2024 | Global Health |
In October 2024 the European Investment Bank (EIB Global) announced €2 million financing for early-stage vaccine development in Rwanda by Akagera Medicines Africa Limited. The support is intended to accelerate research, development, and manufacturing of new vaccines against infectious diseases like tuberculosis, HIV, Lassa fever, and Ebola. It will also be used to “strengthen technical skills and expertise” to support “home-grown discovery, manufacturing, and development of vaccine delivery systems” in Rwanda.
Global Gateway
This financing is part of the EU Global Gateway initiative, a strategy to “boost smart, clean, and secure links in digital, energy, and transport sectors and to strengthen health, education, and research systems”. Team Europe is mobilising up to €300 million between 2021 and 2027 to “allow EU’s partners to develop their societies and economies” whilst creating opportunities for EU Member States to “invest and remain competitive”. EIB Global supports “high impact investment” to enhance healthcare and pharmaceutical manufacturing, encourage greater “health resilience”, and support equitable access to healthcare.
Continent-based solutions
EIB Global states that Africa bears the highest disease burden globally, demanding “more home-grown or continent-based solutions”.
“Vaccination is a critical activity to ensure and guide investments in universal health and has a crucial role to play in achieving 14 of the 17 United Nations Sustainable Development Goals.”
Akagera Medicines was founded in 2018 and registered a 100%-owned subsidiary in Kigali in 2022. Its mission is “targeting tuberculosis and other infectious diseases with liposomal nanotherapeutics”. Commenting on the financing announcement at the World Health Summit in Berlin, Chief Executive Officer Michael Fairbanks recognised the “significant support” of the European Investment Bank.
“We are now a clinical company and moving faster to build human capacity and specialised infrastructure in Africa to support vaccine development.”
CEO of the Rwanda Social Security Board (RSSB) Regis Rugemanshuro stated that the financial support is an “important contribution to the realisation of Rwanda’s vision to become a biotech hub” and the wider vision of “Africa becoming self-reliant in vaccine and medicine manufacturing”.
“RSSB is looking forward to deepening partnerships with EIB and other international institutions to build resilient healthcare ecosystems in Rwanda and in Africa.”
Vice President of EIB Thomas Ostros identified the Bank’s “close cooperation with public and private partners” to “accelerate development of innovative solutions”.
“The EIB is committed to further strengthening our partnership with local and international players, to scale up investment and support innovative technology together.”
Belen Calvo Uyarra, EU Ambassador to Rwanda, agreed that the investment was another “important milestone”.
“Through Global Gateway, the EU is focussed on advancing equitable access to health products and local manufacturing in Africa.”
For more from key players in efforts to establish local manufacturing capacities in Africa and champions of equitable access to health products, join us at the Congress in Barcelona later this month. Don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 11, 2024 | Global Health |
A WHO report in October 2024 suggests that vaccines against 24 pathogens could reduce the number of antibiotics needed by 22% every year. Some of these vaccines are already available but currently underused, but others will need to be developed and brought to market. The report expands on a WHO study from 2023, estimating that some vaccines already in use could avert up to 106,000 deaths caused by AMR each year. Director-General Dr Tedros Adhanom Ghebreyesus highlighted that addressing AMR “starts with preventing infections”, for which vaccines are “among the most powerful tools”.
“Prevention is better than cure and increasing access to existing vaccines and developing new ones for critical diseases, like tuberculosis, is critical to saving lives and turning the tide on AMR.”
The burden of AMR
Antimicrobial resistance (AMR) is the result of bacteria, viruses, fungi, and parasites changing to stop responding to medicines. As medicines become ineffective, infections become harder to treat, which increases the risk of disease spread, severe illness, disability, and death. The report introduces the significant global burden of AMR. In 2019, an estimated 7.7 million deaths were associated with 33 bacterial infections, with almost 5 million of these associated with AMR.
The mortality burden of these drug-resistant infections is “most pronounced” on the African continent, followed by South-East Asia and Eastern Europe. However, community mobility increases the risk of transmission to other continents. AMR has the potential to impose an annual global cost of up US$3.4 trillion by 2030, with the most severe consequences for low- and middle-income countries (LMICs).
A “key driver’ of AMR is the “systematic misuse and overuse” of antimicrobials in healthcare, animal health, and agriculture; the greatest contributor to overall use of antimicrobials is use in animals. The World Organisation for Animal Health (WOAH) estimated that 84,500 tonnes of antimicrobials were used in the animal sector in 2019. However, this is a 13% decrease from 2017. On the other hand, global antibiotic consumption in humans increase by 65% between 2000 and 2015 and is projected to triple (from 2015) by 2030.
One of the major challenges is ensuring equitable access to antimicrobials, particularly in LMICs, where “people are more at risk of dying from a lack of access to appropriate antimicrobials than from resistant infections”. Managing AMR demands both sector-specific and “One Health” approaches. Vaccines can be critical to efforts to lower the burden by reducing the incidence of drug-sensitive and drug-resistant infections, antibiotic use, and opportunities for evolution and transmission of resistant genes and pathogens.
The report
Although we know that vaccines are important aspects of the solution, their specific role in reducing AMR has not been “systematically evaluated and quantified”. Therefore, the latest report evaluates this and provides recommendations for “enhancing the impact of vaccines on AMR”. It covers 44 vaccines targeting 24 pathogens, drawing the characteristics of each vaccine from various sources. Three criteria were considered:
- The AMR-related health burden – measured by the reduction in deaths and DALYs associated with AMR
- Antibiotic use (or antimicrobial use in the case of Mycobacterium tuberculosis)
- The economic burden of AMR, including hospital costs and productivity losses
Highlights from the report
- Vaccines against 16 bacterial pathogens may prevent 510,000 deaths and 28 million DALYs associated with AMR.
- This prediction increases to include an additional 1.2 million deaths and 37 million DALYs when the use of vaccines is expanded to target all populations at risk of infection.
- The non-serotypespecific vaccine against S. pneumoniae, with increased efficacy against lower respiratory tract infections, would have the highest impact on both AMR-associated deaths and DALYs.
- The greatest impact of vaccines on reducing the burden of bacterial AMR in 2019 was in the WHO African Region, averting an estimated 170,000 deaths and 12 million DALYs annually.
- In the WHO South-East Asia Region, vaccines were estimated to have prevented 160,000 deaths and 7.5 million DALYs annually.
- The development and optimal use of vaccines against 23 pathogens could avert up to 2.5 billion defined daily doses a year, which is 22% of the global estimated antibiotic use in humans associated with treating these pathogens.
What’s next?
The authors suggest that the role of vaccines in addressing AMR is “often overlooked” in policy and decision-making processes. They highlight the need for “greater recognition and integration” of vaccines into AMR mitigation strategies and the importance of considering AMR in vaccine decision-making.
“To achieve appropriate inclusion of vaccines in the AMR agenda, the immunisation and AMR communities must strengthen their joint understanding of the evidence and enhance collaboration.”
How do you think that AMR priorities can be incorporated into vaccine development and deployment efforts? Join us for the AMR and bacterial vaccines track at the Congress in Barcelona this month to contribute to these conversations, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 10, 2024 | Global Health |
In October 2024, WHO announced an agreement with the International Monetary Fund (IMF) and the World Bank Group (WBG) on “broad principles for cooperation on pandemic preparedness”. The cooperation is intended to enable scaling up of support to countries to prevent, detect, and respond to public health threats through the IMF’s Resilience and Sustainably Trust (RST), WBG’s financial and technical support, and WHO’s technical expertise and in-country capabilities. The organisations will “leverage their experience to enhance pandemic preparedness”, working on the “synergies and complementarity” of each institution’s in-country analysis and operations.
Principles of coordination
Under the Broad Principles of Coordination:
- WHO and the WBG will continue to lead on health-related development policies and, with other multilateral development banks and The Pandemic Fund, on specific project investments for pandemic preparedness. RST financing will not be earmarked for specific projects.
- Pandemic preparedness policy reform measures supported by RSF arrangements will be informed by existing data, analytics, and operational engagement of WHO, the WBG, and country authorities.
- Pandemic preparedness reforms will build on each institution’s area of expertise. RSF programmes will focus on macro-critical policy reforms within the IMF’s expertise and complement the work carried out by the WBG and WHO to maximise both the financial resources and technical expertise available to countries. RSF Reform measures can include policy actions aimed at enhancing the readiness of finance and health systems to respond effectively to future health emergencies.
The cooperation will enable all three institutions to better serve countries’ efforts on pandemic preparedness.
Working for a safer world
Kristalina Georgieva, Managing Director of the IMF commented that the “stepped-up collaboration” will help the organisations to “complement and leverage each other’s expertise” to support members’ pandemic preparedness and resilience efforts.
“The IMF’s Resilience and Sustainability Trust allows eligible member countries to access affordable, long-term financing to address structural challenges that threaten their macroeconomic stability.”
WHO Director-General Dr Tedros Adhanom Ghebreyesus reflected on the need for “new sources of financing to bolster health systems”, making them “more able to prevent and detect” health threats and to “respond and withstand them when they strike”.
“WHO is proud to be working with the IMF and the World Bank to unlock financing from the Resilience and Sustainability Trust, and support countries to put it to work for a safe world.”
World Bank Group President Ajay Banga suggested that the “deepened collaboration” will focus efforts to help countries prepare for and respond to health threats.
“We must aggressively be planning and preparing for the next global health crisis, so that when the battle comes – and we know it will – we will have the health workforce that can be rapidly deployed in the face of a crisis, laboratories that can quickly ramp up testing, and surge capacity that can be called upon to respond.”
For insights into pandemic preparedness initiatives at the Congress in Barcelona this month get your tickets here, and don’t forget to subscribe to our weekly newsletters here!

by Charlotte Kilpatrick | Oct 10, 2024 | Global Health |
The African Union Development Agency – New Partnership for Africa’s Development (AUDA-NEPAD) announced in October 2024 that Wellcome is granting US$12,301,075 to “support the strengthening and harmonisation of regulatory systems and the operationalisation” of the African Medicines Agency (AMA). The grant will help to drive efforts to “overcome regulatory capacity challenges” to improve access to essential medical products and technologies.
The African Medicines Regulatory Harmonisation (AMRH) initiative has been “pivotal” at creating a “cohesive regulatory environment” for the pharmaceutical sector across Africa since 2009. It focuses on using Regional Economic Communities (RECs) to ensure that African populations have access to high-quality, safe, and effective medical products and health technologies. Wellcome’s grant will be used in alignment with AMRH’s vision of “overcoming barriers” like “limited human and institutional capacity, fragmented regulatory processes, and inconsistent technical standards”.
AMA
AMA’s vision is that “African people have access to essential medical products and technologies”; it hopes to achieve this through the mission: “provide leadership in creating an enabling regulatory environment for pharmaceutical sector development in Africa”. The funding is expected to accelerate efforts to create a “unified and efficient” regulatory framework. The partnership between Wellcome and AUDA-NEPAD is a “major advancement in the pursuit of a robust and harmonised regulatory environment” with positive effects for health outcomes in Africa.
Efficient, connected, fair
Symerre Grey-Johnson Director for Human Capital and Institutional Development at AUDA-NEPAD, stated that the “generous support” from Wellcome is a “crucial endorsement of our mission”.
“With the African Medicines Regulatory Harmonisation (AMRH) intiative laying the groundwork for the African Medicines Agency (AMA), this grant will empower us to address significant regulatory challenges and enhance access to essential medical products for millions of Africans.”
Mr Grey-Johnson believes that the collaboration will “solidify the foundation of the AMA” and ensure a “robust and harmonised” regulatory environment across the continent. Dr Sally Nicholas, Wellcome’s Head of Health Systems and Environment, recognised the AMA’s “crucial role” in creating a “more efficient, connected, and fair regulatory system” in Africa.
“Strengthening regulatory systems is fundamental to improving healthcare outcomes for Africa. By supporting innovative partnerships, initiatives, and solutions to help coordinate effectively operationalise the AMA, we can ensure equitable access to much-needed vaccines, treatments, and interventions for those with the greatest need.”
At the Congress in Barcelona this month we look forward to learning about an AMA pilot with MSD in the Supply and Logistics track; get your tickets to join us there and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 30, 2024 | Infection |
In September 2024, WHO’s Regional Office for Africa announced that Rwandan health authorities are “intensifying outbreak control efforts” after detecting Marburg virus disease in the country for the first time. 26 cases have been confirmed in seven of the country’s districts, and six deaths have been reported. The health authorities are implementing “comprehensive response measures” and an investigation to determine the origin of the outbreak. WHO states that it is supporting these efforts with expertise and tools.
Cases reported and response triggered
26 cases have been reported; 20 are in isolation and receiving treatment, and six deaths have been recorded. 161 contacts of the reported cases have been identified so far and are being monitored. The Ministry of Health, Rwanda, posted a video on social media with a caption reassuring viewers that “people can continue with their daily activities” and “should not panic” as the “hotspots of the disease” have been identified.
WHO is “mobilising” expertise and outbreak response tools to “reinforce the control measures” that are being rolled out. A consignment of clinical care and infection prevention and control supplies will be delivered from the WHO Emergency Response Hub in Nairobi, Kenya, to Kigali in the next few days. Efforts are also underway to “reinforce collaborative cross-border measures for readiness and response” in countries that neighbour Rwanda.
WHO Regional Director for Africa, Dr Matshidiso Moeti, explained that the critical outbreak response aspects are being put in motion “rapidly” to “halt the spread of this virus swiftly and effectively”.
“With the country’s already robust public health emergency response system, WHO is collaborating closely with the national authorities to provide the needed support to further enhance the ongoing efforts.”
Marburg
Marburg virus disease is a “highly virulent” member of the filoviridae family and causes haemorrhagic fever. It has a fatality ratio of up to 88%, with symptoms progressing rapidly after infection. The virus is transmitted to humans from fruit bats and spread among people through direct contact with the bodily fluids of infected people, surfaces, and materials.
The disease was first recognised after large outbreaks in Germany and Serbia in 1967, associated with laboratory research involving African green monkeys from Uganda. Outbreaks and cases have been reported sporadically since then, and efforts have been made to develop effective medical countermeasures. However, there is no licensed vaccine against Marburg virus disease.
A confluence of infectious disease
The Marburg outbreak will increase pressure on the Rwandan health system, which is already fighting its mpox outbreak, declared on 27th July 2024. In September 2024, Dr Jean Kaseya, Director-General of Africa CDC, reported that Rwanda had begun an mpox vaccine campaign after receipt of 1,000 doses. The campaign targeted districts bordering the Democratic Republic of the Congo, the epicentre of the current PHEIC. How will the health services respond effectively to both infectious disease threats, and will there be similarities or ‘doubling up’ in their strategies?
For the latest infectious disease and vaccine news, don’t forget to subscribe to our weekly newsletters here. To participate in important discussions about vaccine development in outbreak situations, join your colleagues at the Congress in Barcelona next month.

by Charlotte Kilpatrick | Sep 25, 2024 | Global Health |
At the United Nations General Assembly High-Level Week in September 2024, CEPI launched a new Biosecurity Strategy to bolster global health security and emphasise its commitment to addressing emerging epidemic threats. The strategy draws on the latest technologies and encourages international collaboration to mitigate the risks presented by emerging pathogens. This is a “significant step” in the “evolution” of CEPI, positioning it as a “though leader in the rapidly developing fields of biosafety and biosecurity”. Chair of CEPI’s Board, Professor Jane Halton, positioned the 100 Days Mission at the centre of this strategy and highlighted the significance of “security and equity” in this effort.
“To underpin that strategy, and to ensure the world can achieve the 100 Days Mission goal safely and securely, we need a robust, collaborative approach to maximising the benefits of new technologies and reducing their potential threats to human health.”
Remaining vigilant and stepping up
Dr Richard Hatchett, CEO of CEPI, writes in the foreword of the importance of remaining “vigilant” and being able to respond quickly to infectious disease outbreaks in an era of “heightened epidemic and pandemic risk”. He reflects that COVID-19 demonstrated the “devastating global consequences” of a pandemic and, through the “persistent controversy” over the origins of the pandemic, heightened awareness of the risk of accidental release and deliberate misuse of science.
“Most risk created by advances in the biological sciences derives from the fungibility of the tools designed to solve specific problems. The tools that will solve a pressing problem are empowering – but there is no intrinsic limit to their application.”
Dr Hatchett acknowledges the problem of “dual use” in the way that biologists tackle problems. However, he warns against imposing limits on scientists, suggesting that this could present “practical challenges” and “impede our progress towards legitimate and worthy goals”, among which is the 100 Days Mission.
“Global scientific participation is critical to the success of the 100 Days Mission and will enable vaccine research, development, and manufacturing to take place in communities that need it, led by those who will benefit from it, and informed by the priorities of the vulnerable communities that are disproportionately impacted by epidemics and pandemics.”
To address the risk of accidents or misuse, Dr Hatchett highlights the importance of mechanisms to “ensure that the highest, most current standards of biosecurity and biosafety are practiced and maintained”. CEPI’s “highly diverse” research portfolio includes more than 50 countries, each with “highly variable” oversight practices. The need for a biosecurity strategy is directed by a recognition that “as a steward of global funds, no matter where those funds are deployed, we have a critical responsibility to ensure that the research we fund does not lead to the next accident or deliberate incident”.
‘Beyond this threshold obligation, CEPI also has an opportunity to step-up as a thought leader in this emerging area.”
In developing the strategy, CEPI engaged more than 150 entities in the global health and security ecosystems in a consultative process and sought advice from a Biosecurity Strategy Group. Although technological capabilities will evolve and “boundaries blur between disciplines”, the strategy anticipates that “traditional approaches” may prove “inadequate” in the face of emerging threats. Thus, stakeholders must collaborate and develop mechanisms to encourage responsible use, supported by CEPI.
Biosafety and biosecurity
The strategy acknowledges the evolution of the terms biosafety and biosecurity, comparing the WHO (2024) definition of biosecurity with FAO’s (2007) understanding of the term. CEPI’s biosecurity and biosafety priorities must align with its mission to accelerate vaccine development towards the 100 Days Mission. The strategy outlines how an “innovative approach…frontloaded towards preparedness”, can enable the safe and secure delivery of this goal.
Top vulnerabilities
Several biosecurity and biosafety vulnerabilities are identified and considered relevant to the strategy:
- Variable biosafety and biosecurity oversight, risk identification, and management practices among life science funders for research involving high consequence pathogens, including CEPI.
- Substantial variations in biosafety and biosecurity policies, regulations, practices, and competencies where CEPI-funded research takes place, and insufficient health and security collaboration.
- The intersection between biosecurity and equity is insufficiently recognised, which threatens progress towards the 100 Days Mission and future responses to epidemic and pandemic threats.
- Emerging biotechnology and converging technologies present dynamic and evolving biosecurity risks that threaten 100 Days Mission progress.
- The world is insufficiently harnessing technological innovation to reduce safety and security vulnerabilities of the 100 Days Mission.
Focus and priorities
CEPI’s biosecurity focus is to “protect society from epidemic and pandemic threats, with an emphasis on preventing accidental and deliberate misuse of pathogens associated with CEPI-sponsored research”. The strategy therefore addresses global biosecurity vulnerabilities to accelerate current strategic goals with the following priorities:
- Strengthen biosafety and biosecurity risk identification, mitigation, and oversight by CEPI and encourage similar efforts by other life science research funders.
- Enhance global biosafety and biosecurity capabilities of CEPI partners for achieving the 100 Days Mission safely and securely and promote health-security partnerships.
- Drive biosecurity and biosafety in support of equity.
- Monitor and reduce emerging biotechnology and converging technology risks across CEPI’s vaccine research, development, and manufacturing portfolio.
- Accelerate biosafety and biosecurity innovation for vaccine research, development, and manufacturing.
More to come
An implementation plan of activities, goals, and timelines will follow the strategy. It will explore how priorities can be integrated into the wider mission and mandate in three major categories:
- Catalysing strategic partnerships and coalitions
- Advocacy and coordination
- Supporting biosafety and biosecurity capabilities development
Director General of Africa CDC, Dr Jean Kaseya, expressed enthusiasm at the strategy launch and its support of Africa CDC’s efforts.
“With a focus on laboratory systems strengthening, training and infrastructure development, and reducing risks of artificial intelligence and other innovations, the strategy is informed by vulnerabilities across a wide range of resource settings and will help galvanise global progress toward safely and securely achieving the 100 Days Mission.”
Trevor Smith, Deputy Director at Global Affairs Canada and member of the CEPI Biosecurity Strategy Group, welcomed the focus on “effective collaboration between the health and security sectors”.
“The strategy articulates an ambitious vision for reducing vulnerabilities and strengthening global health security.”
For insights from senior representatives of CEPI at the Congress in Barcelona next month, get your tickets to join us here. Don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Aug 28, 2024 | Global Health |
Research in Nature Medicine in August 2024 presents the importance of a safe and effective Lassa vaccine deployed across 15 countries of continental West Africa. The authors find that vaccination against Lassa fever could save nearly 3,300 lives over 10 years and avert up to $128 million in societal costs. Alongside estimating the health-economic burden of Lassa fever in West Africa, the study models the emergence of ‘Lassa-X’, a hypothetical pandemic Lassa virus variant, and projects the effects of achieving 100 Days Mission vaccination goals.
Lassa fever
Lassa fever, a viral haemorrhagic disease that is endemic to West Africa, is caused by Lassa mammarenavirus (LASV). Although infections are common but “widely undetected”, it is believed that most human LASV infections are caused by zoonotic transmission from the Natal multimammate mouse (Mastomys natalensis). It can also spread through human-to-human contact, largely in healthcare settings with “inadequate infection prevention and control practices”.
Most LASV infections are asymptomatic or cause mild febrile illness, but Lassa fever has a “large negative impact” on population health and economies. Among patients who present to hospital, the case-fatality ratio is around 15%. Long-term sequalae, including bilateral sensorineural hearing loss, are common in survivors. Costs per hospitalisation are “high” and often (at least partly) paid out of pocket by patients. There are no licensed vaccines against Lassa fever, but several candidates are in development. Lassa fever is considered a threat by WHO because it has epidemic potential and an absence of effective countermeasures.
The study
The authors estimated the current health-economic burden of Lassa fever in West Africa and project the possible effects of different reactive and preventive vaccination campaigns. They also project the potential effects of vaccination in line with the 100 Days Mission in response to a hypothetical future variant of LASV with pandemic potential. Their epidemiological model project the human Lassa fever burden over 10 years in 15 countries: Benin, Burkina Faso, Côte d’Ivoire, The Gambia, Ghana, Guinea, Guinea-Bissau, Liberia, Mali, Mauritania, Niger, Nigeria, Senegal, Sierra Leone, and Togo. These countries had 183 level 1 administrative units, known in the study as “districts”.
“Due to large gaps in Lassa fever surveillance and limited case reporting throughout much of its endemic range, we favoured a bottom-up modelling approach, synthesising best available ecological, epidemiological, clinical, and economic data to project the cumulative health and economic burden of disease.”
The model comprised six main components:
- A previously published geospatial risk map was used to predict the risk of spillover at the level of 0.05° x 0.05° spatial pixels throughout West Africa.
- Modelled spillover risk estimates were used as inputs in a generalised linear model (GLM) to predict human LASV seroprevalence.
- Modelled human LASV seroprevalence estimates were used as inputs in a serocatalytic model including country-level population projections to predict spillover infection incidence.
- Spillover infections were aggregated at district level and a stochastic branching process model was used to simulate onward human-to-human LASV transmission.
- A computational algorithm was applied retrospectively to spillover infections and ensuing transmission chains to simulate a range of reactive and preventive vaccination campaigns and to project the number of infections averted by vaccination.
- Modelled estimates of LASV infection and infections averted through vaccination strategies were used as inputs in a probabilistic decision-analytic model used to project the health burden of Lassa fever and associated economic costs and the health and economic burden averted due to vaccination over 10 years.
Vaccination
Vaccination was introduced in a series of six scenarios that reflected “realistic assumptions” about vaccine stockpile, administration, and efficacy. Every scenario included reactive vaccination, in which outbreaks trigger the local deployment of a limited vaccine stockpile in affected districts. The authors considered two main mechanisms of vaccine efficacy:
- Protection against infection prevents individuals from acquiring LASV infection from either M. natalensis or other humans
- Protection against disease prevents vaccinated individuals who become infected from progressing to disease, thus averting outpatient consultation, hospitalisation, chronic sequalae, and death
In the simulations, the researchers’ projections feature a vaccine that is 70% of 90% effective only against disease or 70% or 90% effective against both infection and disease.
Lassa-X
The paper also presents modelling of the emergence of “Lassa-X”, assumed to emerge in humans after a single spillover event. Prior LASV immunity was assumed to offer no protection against Lassa-X. Lassa-X was conceptualised with “Ebola-like transmission characteristics” and a 10-fold increase in hospitalisation risk relative to Lassa fever. Vaccination against Lassa-X was also considered; the “most ambitious” vaccination scenario achieved the 100 Days Mission of administration 100 days after initial detection of the first hospitalised case.
Findings
The researchers estimate that 2.1-3.4 million human LASV infections occur each year in West Africa, resulting in 15,000-35,000 hospitalisations and 1,300-8,300 deaths. Lassa fever was estimated to cause 2.0 million disability-adjusted life years (DALYs), $1.6 billion in societal costs, and $15.3 billion in lost value of statistical life (VSL) over 10 years.
The modelling suggests that administering Lassa vaccines preventively to districts classed as endemic in Nigeria, Guinea, Liberia, and Sierra Leone would avert a “substantial share of the burden of disease in those areas”. In the most expansive rollout scenario, in which a vaccine reaches around 80% of individuals in endemic districts and 5% of individuals elsewhere over 3 years, a vaccine that is 70% effective against disease is projected to avert 164,000 DALYs, $128 million in societal costs, and $1.3 billion in VSL lost over 10 years. For the same scenario, a vaccine that is 90% effective against both infection and disease could avert 240,000 DALYs, $188 million in societal costs, and $1.9 billion in VSL lost.
Vaccination campaigns in the other countries in the analysis had “modest” effects due to a reflection of a “constrained global vaccine stockpile”, meaning limited allocation to non-endemic districts.
“It is important to put Lassa fever’s projected health-economic burden and impacts of vaccination in context, in particular given limited economic resources available for investment in infectious disease prevention in West Africa and, hence, opportunity costs to investing in Lassa vaccination in lieu of other interventions.”
Real-world cost-effectiveness of a Lassa vaccine would depend on dosage, price, and clinical efficacy as well as the alternative interventions that are available. For example, novel small-molecule antivirals and monoclonal antibodies may be “promising alternatives” for prevention of severe disease. However, investment in Lassa vaccination has the “major potential benefit” of “increased readiness” for the rapid development and deployment of vaccines against future variants with pandemic potential.
Conclusions and comments
The authors conclude that vaccination campaigns that target “known Lassa fever hotspots” will help to reduce the large health-economic burden. However, it will be important to expand vaccination beyond WHO-classified endemic districts. Improved surveillance is also “greatly needed”, particularly to inform vaccination campaigns. Finally, in the hypothetical event of a novel, highly pathogenic pandemic variant “emerging and devastating the region”, the study suggests that the 100 Days Mission vaccination targets could have “critical impact”.
“The probability of such a variant evolving is exceedingly difficult to predict, but investment in Lassa vaccination now could nonetheless have great additional health-economic value if facilitating a more rapid vaccine response in the event of a pandemic Lassa-related virus emerging.”
CEPI’s CEO Dr Richard Hatchett warned that Lassa fever, a “serious public health problem in West Africa”, is already likely to spread to other regions as “climate and environmental change increase epidemic risk”.
“This study demonstrates the urgent need for a vaccine to protect people from this debilitating and sometimes deadly disease which we believe affects many more than those who are reported, due to limited access to diagnostics and healthcare.”
Lassa fever remains a priority for CEPI, and Dr Hatchett is “proud” that CEPI is a world leading Lassa vaccine R&D funder. Dr Virgil Lokossou, Head of Division, Preparedness and Response at the West African Health Organisation, reflected on the “burden” and “significant socioeconomic consequences” highlighted in the paper, revealing the “urgent need to accelerate vaccine research and development”.
“The West African Health Organisation remains committed to working with our Member States, CEPI, and all stakeholders to ensure that we fast-track the development of a vaccine and other tools we need to control the spread of Lassa fever and protect our communities. Time is now up for concrete actions.”
Dr David Smith, Senior Researcher at Oxford Population Health’s Health Economics Research Centre, joint first author, called for investment in Lassa vaccination.
“One major potential benefit of present investment in Lassa vaccination development is increased readiness to rapidly develop and deploy vaccines against future Lassa variants with pandemic potential.”
Dr Joanne Turner, research associate at the University of Liverpool, joint first author, shared that the analysis included vaccination campaigns designed to reflect “realistic assumptions”.
“Consequently, the impacts of our simulated Lassa vaccination campaigns were modest in countries other than Nigeria, Guinea, Liberia, and Sierra Leone. Yet the data underlying our model suggest that there is likely already a significant burden of Lassa fever outside these countries.”
Professor of Infectious Disease Epidemiology at the University of Oxford Big Data Institute, Déidre Hollingsworth, emphasised the importance of a vaccine to key populations.
“Lassa fever predominantly affects low-income populations in rural areas and is likely to be underreported due to poor health access in these areas.”
We will hear more about a Lassa fever vaccine candidate and the “challenges” of a field efficacy study in West Africa from IAVI’s Dr Marion Gruber at the Congress in Barcelona this October. Get your tickets to join us there, and don’t forget to subscribe for weekly vaccine updates.

by Charlotte Kilpatrick | Aug 19, 2024 | Global Health |
The Aga Khan Foundation (AKF) announced in August 2024 that it is launching a $7.2 million nutrition and immunisation programme in Pakistan with support from federal and provincial governments, Gavi, and The Power of Nutrition (TPoN). The programme seeks to support more than one million mothers and children in the most marginalised areas of three provinces. Pakistan faces “significant” child health challenges, with the third-highest global burden of child mortality; it ranks third in the world for the “most under-vaccinated children” with nearly 1.2 million children not immunised. In hard-to-reach populations, where there are higher concentrations of “undernourished, stunted, and wasted children”, there are high numbers of “zero-dose” children.
Malnutrition and under-immunisation
AKF infers from the correlation of malnourished and under-immunised children that children who are at high risk of malnutrition are also the ones missing out on essential immunisation services. Therefore, an integrated immunisation and nutrition approach could provide “combined reinforcement benefits”. Although malnutrition and infectious diseases are “key contributors” to child morbidity and mortality, immunisation and nutrition programmes “often operate in isolation”.
$7.2 million programme
The programme will support districts with a “particularly high” need: Diamir, Astore, Gilgit, Sibi, Bolan, Usta Muhammad, Thatta, and Sajawal. It is jointly funded by the partners and centrally managed by The Power of Nutrition. Starting later this year, it will run until 2027.
The integrated approach involves strengthening health systems to address existing gaps, social behaviour change communication to ensure demand for immunisation, and support for district and national governments towards evidence-based decision making and learning. It seeks to provide “vital evidence” on the importance of integration and real-life examples to demonstrate cost-effective methods of joint delivery.
Akhtar Iqbal, Chief Executive Officer of Aga Khan Foundation Pakistan, looks forward to the “unique opportunity” to contribute to Sustainable Development Goals by “extending an integrated package of immunisation, health, and nutrition interventions for children living in some of the most marginalised districts in Pakistan”.
“Through a close partnership with the Federal and Provincial Expanded Programme on Immunisation Directorates, and technical support of the Aga Khan Health Services and Aga Khan University, the programme will generate data, evidence, and learning to fill gaps and discover what works in this under-resourced area.”
Dr Tokunbo Oshin, Director, High Impact Countries, Gavi, is “pleased to be able to support this innovative programme”, which addresses parental preferences to be “reached with package of interventions”.
“Through health systems strengthening efforts, this will be a good opportunity to provide essential services in remote areas of Pakistan and learn how to better scale up integrated service delivery, including immunisation and nutrition.”
Dr Alok Ranjan, Director of Programmes and Investments, The Power of Nutrition, is “delighted to bring together” the partners for a “vital project”.
“For too long nutrition and immunisation stakeholders have been working separately, despite the interventions reaching similar populations and being mutually beneficial. This programme promises not only real impact in Pakistan, [but] it can help pave the way for more integrated programming worldwide.”
To hear from immunisation experts at the Congress in Barcelona this October get your tickets here and don’t forget to subscribe to our weekly newsletters for vaccine updates.

by Charlotte Kilpatrick | Jul 16, 2024 | Global Health |
In July 2024, Grattan Institute shared a report on the importance of GPs in providing vaccination services. The authors, Peter Breadon and Anika Stobart, call for better government support for these “trusted experts” in delivering this service, such as increased investment or better data. Although there are other barriers to vaccination, Breadon and Stobart describe the role of GPs in closing the vaccination gap as “clearly crucial”. Their report presents specific recommendations to improve vaccination rates for GP patients, suggesting that the government should “level the playing field”.
Differing vaccination rates
“GPs are the foundation of Australia’s healthcare system. As well as diagnosing and treating many conditions, GPs give advice and treatment to prevent problems or stop them getting worse. This includes ensuring patients are up to date with their recommended vaccinations.”
The report begins by identifying a range in GP patient vaccination rates, with some GPs having “very low” adult vaccination rates and others exceeding expectations. These rates range from a two-fold difference for flu to a 13-fold difference for pneumococcal. The lowest coverage is linked to “disadvantaged areas”, “culturally and linguistically diverse backgrounds”, and “residential aged care”. This is concerning as these groups are “often” at “greater risk of severe illness”.
“Structural barriers to vaccination are often to blame for low vaccination, not individual, or cultural, attitudes towards vaccines.”
To determine variation in adult vaccination rates and which GPs might need “more targeted support”, the authors used data at patient and GP level. For COVID vaccination, the bottom 5% of GPs (around 1,600) had only 16% of their patients aged 65 and older up to date, less than one third of the average. Flu vaccination saw a two-fold difference, with GPs with lowest flu coverage having only 43% of their patients vaccinated compared to 90% for highest flu coverage.
Shingles had a four-fold difference, ranging from 5% to 22% on average; the top 5% had 85% of their patients up to date. However, pneumococcal had the “biggest disparity”. The bottom 5% of GPs had only 6% of their patients vaccinated, compared to the top 5% who had 82% of their patients vaccinated. This was a 13-fold difference.
“While some variation is inevitable, these differences are unacceptably large, especially for COVID and pneumococcal. They suggest the system is failing to give everyone good access to potentially life-saving preventative healthcare.”
Key barriers
The report identifies an “imbalance” in the health system, finding that “Australians who are more vulnerable to disease are less likely to get the high-quality, preventive care they need”. Factors such as proficiency in English, where someone lives, or highest level of education attained, have a “big effect”. For example, people who speak a language other than English at home or “do not speak English proficiently”, are “much less likely” to be up to date with vaccinations. People born in North Africa and the Middle East were between 15 and 25% less likely to be vaccinated than people born in Australia, and being born overseas was “nearly always associated with lower vaccination”.
“These low vaccination rates could be due to a range of factors including discrimination, language barriers, lower health literacy, or higher rates of vaccine hesitancy. But because vaccination is lower for virtually all non-English language groups, and for migrants from almost all international regions, it is clear that the problem is not just about the beliefs or preferences of any specific cultural group.”
Other groups are also less likely to be vaccinated, including “older people with higher rates of economic disadvantage” and people with “lower levels of education”. Although people from “disadvantaged areas” suffer greater rates of chronic disease and are “more than twice as likely to die from potentially preventable causes”, people in “outer regional and remote areas” are less likely to be vaccinated.
“Groups who need healthcare more having a greater risk of missing out is a systemic problem that goes well beyond vaccination”.
People in aged care are also at “significant risk” of severe illness and death from disease, but their vaccination rates are “far too low”. For example, only 38% of aged care residents were up to date with COVID vaccinations in June 2024. Almost 100 aged care facilitates had less than 10% of residents vaccinated against COVID in a year, and a quarter of facilities have “less than half” of their residents vaccinated. These rates are “far below” those of “similar countries” – the authors refer to Ireland and England as examples.
Another trend is that people who visit a GP “frequently, regularly, or who visit more expensive GPs, are more likely to be vaccinated”. People who visit a GP “frequently” are up to 27% more likely to be vaccinated than those who don’t, and seeing a GP who charges more “increases your chances of being vaccinated” by up to 17%. While the report highlights a “range of barriers” to vaccination and recognises that they demand “structural changes to healthcare”, it suggests that “some” differences in GP vaccination rates could be attributed to differences in GP practices.
GP practices
Despite accounting for a “wide range” of barriers to vaccination, the report found that some GPs still have lower rates than might be expected. The authors infer that “other factors” may be contributing. These might include the effects of the primary care funding model, which has caused GPs to compress “increasingly complex care” into appointments that average around 15 minutes. They are “under pressure” to complete appointments quickly and “skip” preventive healthcare. Therefore, they may find it hard to make time for patients who are hesitant about vaccination, call in interpreters, or handover to a nurse (if there is one), to enable patients to get vaccinated.
Funding and regulatory barriers also mean that GPs might have “less support from a broader team”, such as nurses or allied health professionals. Thus, explaining vaccine risks and benefits or countering misinformation “often falls on the GP alone”. They also might have “different levels of focus” on vaccination; an Australian study found that GPs only initiate discussions about vaccinations in around 10% of consultations. There is also the possibility that GPs are “unaware of the latest vaccination information” as the guidance is “unclear and changing”. Finally, they may not “immediately” have information on a patient’s vaccination history on their software programmes.
What should be done?
“Australia’s health system can only get better and fairer if governments measure care and outcomes and strive to improve them at every level, using funding, data, and focused improvement support.”
A key concern for the authors is that GP clinics should be able to “choose a funding model” that “supports team care” and enables GPs to spend time with complex patients by combining appointment fees with a budget that is based on each patient’s level of need. This “blended” funding model is “common in other countries” and has been introduced by the federal government for some patients through “MyMedicare”. It should be expanded to other patients to increase funding for “older, poorer, and sicker patients”.
“This funding based on need not speed would give GPs the opportunity to provide preventive care to the patients who need it most.”
GPs should also be supported with “better information” on how their patient vaccination rates compare to their peers. The federal government could give Primary Health Networks data to share with GPs, even extending to pharmacies. As Primary Health Networks are “responsible for improving primary care”, they should also get new funding to “expand their work supporting GP clinics to boost vaccination”.
The authors emphasise that efforts to improve vaccination rates “cannot be one-size-fits-all” but should respond to different barriers with specific support. Breadon comments that “Australia urgently needs to lift its vaccination game”.
“Our report shows that the system is failing to give every Australian good access to potentially life-saving preventive healthcare.”
For more on the importance of adult vaccination strategies and approaches to encouraging uptake, get your tickets to join us at the Congress in Barcelona this October, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Jun 28, 2024 | Global Health |
In June 2024 WHO announced that the Child Health and Wellbeing Dashboard has been updated to include the latest data, downloadable datasets, and a more user-friendly interface. Initially launched in collaboration with UNICEF and the Children in All Policies (CAP-2030) initiative, the dashboard is intended to provide policymakers with a data-based tool to inform health programmes, policy development, and evaluation. The dashboard covers issues from childhood survival to educational attainment and should enable policymakers and the public to “track progress”.
Improving health and survival
WHO indicates that 4.9 million children under the age of 5 die every year; nearly half of these children are babies in the first month of their lives. It warns that, according to current trends, 59 countries are set to miss the UN Sustainable Development Goal (SDG) target for under-5 deaths.
“Data is critical for these efforts, helping countries monitor impacts of programmes and policies and guiding interventions to address gaps.”
Staying accountable
The Lancet commentary from 2022 states that the WHO-UNICEF-Lancet Commission on child health and wellbeing called for “renewed commitment” to the “fundamental rights” of children, as enshrined in the UN Convention on the Rights of the Child (CRC). The Commission’s report and subsequent Comment on pandemic “underlined the uncertainties” that children face. It also made recommendations for building a better future for children by centring them in global, regional, and national development agendas and holding governments accountable through a robust cycle of monitoring, reviewing, and acting.
“This cycle depends on the availability of an accountability mechanism that showcases country performance across the four dimensions of the CRC: children’s right to be healthy, protected, educated, and fairly treated and heard.”
The creation of a child flourishing and futures composite index revealed that wealthier countries perform better than poorer countries on child health and development outcomes but are “imperilling children’s futures” through “excessive greenhouse gas emissions and industry practices that are contributing to environmental degradation”. Thus, the Commissioners proposed an additional accountability mechanism to allow countries to monitor progress and act accordingly.
WHO and UNICEF led a consultative process to create a “simple, innovative dashboard” drawing from existing frameworks. In establishing the “basic parameters” of the dashboard, the following steps were agreed:
- A scorecard template would be used with a traffic light style classification system for indicator data.
- The four domain areas of the CRC would be used and labelled as survive, protection, development, and participation, alongside the domain of contextual and policy factors.
- Standard age categories recommended by WHO for children and adolescents would be used.
To select indicators to populate the dashboard, partners combined prioritising indicators in the UN Sustainable Development Goal (SDG) Framework and other frameworks for which data are regularly collected. Then, the thresholds for assessing progress were established, considering the distribution of indicator data values and existing targets.
The updated Dashboard
The Dashboard presents data from 196 countries and territories around the world, providing country-specific data and allowing for global analysis. For example, the indicator that the highest number of countries (144) are struggling with is maternity protections, which WHO suggests are “essential” for supporting both mental and physical health during pregnancy and after birth. The latest version responds to requests from policymakers, academics, and civil society stakeholders during a Town Hall Event in May 2023.
Child and maternal health concerns return to the Agenda for The World Vaccine Congress in Barcelona this October, so do make sure you get your tickets to join us there and subscribe for more global health updates.

by Charlotte Kilpatrick | Jun 3, 2024 | Global Health |
WHO shared in June 2024 that at the annual meeting of its 194 member countries, the World Health Assembly (WHA), a “historic” agreement was reached on “critical amendments” to the International Health Regulations (2005) (IHR) and negotiations on the global pandemic agreement. These decisions were announced on the final day of the 77th WHA and “build on lessons” from several global health emergencies, including the COVID-19 pandemic.
“These critical actions have been taken to protect the health and safety of all people everywhere from the risk of future outbreaks and pandemics.”
Amendments to the IHR
The latest amendments to the IHR include:
- The introduction of a definition of a pandemic emergency to ensure more effective international collaboration in response to events that are at risk of becoming, or have become, a pandemic. This represents a higher level alarm that builds on the existing mechanisms of the IHR, including the determination of a public health emergency of international concern. The definition determines that a pandemic emergency is a communicable disease that has, or is at high risk of having, wide geographical spread to and within multiple States, exceeds or is at high risk of exceeding the capacity of health systems to respond in those States, causes, or is at high risk of causing, substantial social and/or economic disruption, including disruption to international traffic and trade, and requires rapid, equitable, and enhanced coordinated international action, with whole-of-government and whole-of-society approaches.
- A commitment to solidarity and equity on strengthening access to medical products and financing. This includes establishing a Coordinating Financial Mechanism to support the identification of, and access to, financing required to “equitably address the needs and priorities of developing countries, including for developing, strengthening, and maintaining core capacities”, and other pandemic emergency prevention, preparedness, and response-related capacities.
- Establishment of the State Parties Committee to facilitate the effective implementation of the amended Regulations. The Committee is to promote and support cooperation among State Parties for the effective implementation of the IHR.
- Creation of National IHR Authorities to improve coordination of the Regulations within and among countries.
WHO’s Director-General Dr Tedros Adhanom Ghebreyesus identified a “common desire by Member States to protect their own people, and the world’s, from the shared risk of public health emergencies and future pandemics.
“The amendments to the International Health Regulations will bolster countries’ ability to detect and respond to future outbreaks and pandemics by strengthening their own national capacities, and coordination between fellow States, on disease surveillance, information sharing, and response. This is built on commitment to equity, an understanding that health threats do not recognise national borders, and that preparedness is a collective endeavour.”
The Director-General hopes that this IHR progress will provide “powerful momentum to complete the Pandemic Agreement”. Dr Ashley Bloomfield of New Zealand, Co-Chair of the Working Group on Amendments to the IHR (WGIHR), is “so proud to be a part of this”.
“The experience of epidemics and pandemics, from Ebola and Zika to COVID-19 and mpox, showed us where we needed better public health surveillance, response, and preparedness mechanisms around the world. Countries knew what had to be done and we did it.”
Fellow Co-Chair of WGIHR, Dr Abdullah Assiri of the Kingdom of Saudi Arabia, agreed that the amendments “strengthen mechanisms for our collective protections and preparedness against outbreak and pandemic emergency risks”.
“Today’s powerful show of global support for stronger Regulations also provides a great boost for the process to negotiate a much-needed international Pandemic Agreement.”
The Pandemic Agreement
WHO Member States also agreed to extend the mandate of the Intergovernmental Negotiating Body to continue negotiations on a Pandemic Agreement as soon as possible. Precious Matsoso of South Africa, Co-Chair of the Pandemic Accord Intergovernmental Negotiating Body (INB) and the Drafting Group on the INB and IHR agenda items at the WHA, suggested that there was a “clear consensus” among the Member States on the “need for a further instrument to help the world better fight a full-blown pandemic”. Fellow INB Co-Chair Roland Driece of the Netherlands celebrated the “great result”.
“We clearly have the will, the purpose, and now the time needed to complete this generational agreement.”
CEPI calls for more effort
Also in June, CEPI issued a statement welcoming the news that WHO Member States will keep up momentum on negotiations and calling on governments to “make every effort” to reach an agreement “at the earliest possible opportunity”. The statement highlighted a need for efforts on several commitments in the process:
- Sustained preparedness investments in R&D and ensuring that government R&D funding agreements include contractual requirements to support equitable access, such as licensing, affordable pricing, technology transfer, information sharing, and equitable allocation.
- Establishment of a network of partnerships for end-to-end collaboration on medical countermeasures development and response and promotion of strategic investments in economically sustainable, geographically distributed vaccine manufacturing facilities with capability for rapid response.
- Establishment of a multilateral system that supports rapid and efficient sharing of samples and data on pathogens with pandemic potential to expedite R&D for medical countermeasures, at all times, as well as equitable access to the outputs of such R&D. CEPI reiterated that real-time access to 20% of pandemic vaccines should be a minimum requirement for equitable access.
CEPI believes that an agreement that “strengthens accountability, transparency, and international collaboration” will be possible. It hopes for an agreement that “combats structural inequity” and drives the “system-wide change needed to respond more quickly and more fairly” to a future pandemic.
“The world was too slow in its response to COVID-19. This cost the lives of millions of people. We cannot afford to repeat the same mistakes.”
For more on the importance of this agreement in pandemic preparedness and response, join us at the Congress in Barcelona later this year, and don’t forget to subscribe to our newsletters here.

by Charlotte Kilpatrick | May 17, 2024 | Global Health |
Our next interview from the Congress in Washington features Merck’s Dr Jules Millogo, who joined us for a panel in the Vaccine Supply and Logistics track: Partnerships between industry and vaccine implementation programmes. Dr Millogo is another of our interview returners, so we were glad that he was happy to join us again to continue the conversation started last year! A public health physician, Dr Millogo currently works with Merck’s Vaccine Division as WHO Liaison.
Industry meets implementation
We first asked Dr Millogo for some insights into his panel on industry and implementation programmes. His main concern, which he believes is shared by others, is that vaccines are “still not reaching where the disease prevalence is highest in the world”, despite the availability of safe and effective vaccines. Dr Millogo’s role, therefore, is to “attract the attention” of company leaders to ensure there are mechanisms to reach all populations, and to work with governments and international organisations to prioritise the introduction of these vaccines. However, he notes that this role is complicated by specific challenges, such as civil wars or other diseases.
“It’s not necessarily that people are not willing; they’re just overwhelmed.”
Thus, Dr Millogo identifies the need to “facilitate” and ensure that “products that are lifesaving reach those who need it”.
Overcoming obstacles
Our next question considers the key obstacles in place to preventing access to essential vaccines. Dr Millogo suggests that “sometimes the challenges are purely logistics”. For example, he refers to his country of birth, in which travelling 50 miles can take 2 or 3 hours by bicycle or motorbike, and is complicated by a lack of electricity or personnel.
“Those infrastructure challenges can be there.”
A further “barrier” is government prioritisation; Dr Millogo considers the 2006 FDA approval of HPV vaccination, which is now available in “most” high-income countries. However, only 26% of eligible girls are vaccinated. Another cause for concern for Dr Millogo is “zero-dose children”, who miss out on essential vaccines, some of which have been around for many years.
“This is not acceptable. This is something that we all should be concerned about, and make sure we address these barriers.”
While there is progress, we “still have a way to go”, says Dr Millogo; diseases such as measles and polio are “still out there”.
“When you look at the world as it is…we are on the same planet but there are still areas of the planet where it’s a different world really.”
Communication is key for the vaccine field
In our previous interview, Dr Millogo highlighted the life-changing power of trust and effective communication. We asked how receptive the community is to this message.
“It’s really a challenge for us as physicians, public health experts, to adopt the language that people can trust and rely on.”
Dr Millogo reflects that it’s “not intuitive”, and shares that, as a graduate of medical school, even he struggled to keep pace with some of his colleagues at the event. In his experience, it has to be simpler.
“You can really explain in very simple terms; you have to be able to explain to a 5-year-old that the disease is bad and the prevention is good…you don’t need to put in so many expert words.”
For Dr Millogo, this is something that “we have to work harder”.
“Be mindful as ‘experts’, as physicians, or as researchers, or academics, that sometimes we are not the best at communicating this information. We can use more tools.”
Why WVC?
As always, we conclude by inviting our experts to share their reasons for joining us at the event. For Dr Millogo, it’s the opportunity to “make connections that you wouldn’t otherwise have”.
“Here you meet other industry partners…and establish connections that can help really to facilitate access to the products.”
For some of the smaller biotechs, visibility can increase at these events, which means people like Dr Millogo can support their efforts to get interventions to the people who need them most.
“At the end of the day, it’s the human connection that we are all trying to work towards a common goal where people don’t die from preventable causes…we shouldn’t see that anymore.”
We hope that it was a productive event for Dr Millogo, and that you enjoy hearing from him again! To learn about another aspect of his work with the Konkourona Alliance Foundation, click here.
For more conversations with our experts from the Congress in April do make sure you subscribe for weekly updates here!

by Charlotte Kilpatrick | May 8, 2024 | Global Health |
In May 2024 the Local Democracy Reporting Service (LDRS) told the BBC that a decrease in flu vaccine uptake is being attributed to vaccine “fatigue”. The LDRS found that fewer than three in five workers at Sherwood Forest Hospitals NHS Foundation Trust received the flu vaccine this winter, with “health bosses” suggesting a “main reason” for the drop is that staff have already had multiple vaccinations. This is a national trend, according to the LDRS.
Lower than average
“The national NHS flu vaccination target is 80%, but only 43% of workers got it.”
Although frontline healthcare and social workers are eligible to get the vaccine through their employer, numbers are dropping. Within the trust are King’s Mill Hospital, Newark Hospital, and Mansfield Community Hospital. Across these hospitals “fewer than 59% of healthcare staff got the jab”. This is a decrease from 62% in 2023.
The Trust is investigating this growing reluctance. Another factor that is being considered is that the past two flu seasons have been “very mild”, which might be causing complacency among health workers.
Accessibility remains a challenge
Director of people Robert Simcox commented that “one of the main reasons” under investigation is “vaccine fatigue”.
“People have had multiple vaccines before and felt unwell, whether it’s groggy, tired, or sore arms.”
Furthermore, accessibility is “also a challenge”, as vaccinations were easier to get during the pandemic.
“We need to increase the opportunities for staff to have it any time of day in their place of work.”
However, Chair of the board is Claire Ward, who stated that “healthcare workers should be more receptive to arguments about the importance of vaccination”.
“We need to communicate the importance, and hopefully it doesn’t take a bad winter to increase the uptake.”
For more on vaccine uptake concerns and trends, don’t forget to subscribe to our newsletters here.

by Charlotte Kilpatrick | May 8, 2024 | Global Health |
Our next conversation from The World Vaccine Congress in Washington is with Economist Impact‘s Latifat Okara, who joined us to Chair the Market Access track and to discuss the “significance of robust health and economic data in tackling antimicrobial resistance” in the AMR Workshop. Latifat is Principal for Health Policy and Insights, leading up all the team’s research work and insights work for health policy.
Robust data
We first consider Latifat’s session on “robust” data, asking what this means and how it can be achieved. Latifat puts this in the context of AMR.
“What this really means in essence is having thorough and fit-for-purpose data for use as we think about tackling antimicrobial resistance.”
Latifat refers to a report from 2023, which considers an economic model to understand health costs. This research highlighted that, while we have about 16.4 million hospital-acquired infections across 6 emerging economies, there are “still a lot of opportunity gaps”. Furthermore, despite the increasing awareness and attention to antimicrobial resistance, we lack data.
“Without the right data, it’s difficult to measure progress, and without measurement then you really can’t do anything about it.”
Latifat states that, in order to “move the needle”, we need to start thinking about generating “quality data” to use as a “powerful tool” in different conversations.
Why this matters for vaccines
Latifat kindly explains her reasons for emphasising the importance of “robust” data at a vaccine event.
“First of all, without data, there can’t be measurement; without measurement, there can’t be action.”
For the vaccine community, identifying gaps and needs requires data that “cuts across” all levels.
“We need to ask the right questions.”
This goes beyond supplying vaccines to the people who need them; Latifat believes it has economic benefits such as expansion into markets and a “whole lot more”.
Why WVC?
As always, we conclude by asking our experts to share their intentions or expectations for the event, unpacking why they joined us. For Latifat, “practical tools” were key.
“When we think about public health in general, there is a lot of theoretical methods and approaches…but I think for me it’s really important to know how do we take those tools and take it to those who actually need.”
Referring to Dr Cohen’s keynote session, Latifat considers how immunisation can be used at country level.
“How can we actually take the lessons from everything that we’ve learnt from the pandemic…how can we put it to good use?”
In her final comment, Latifat emphasises the importance of breaking the inevitable public health siloes and fostering collaboration.
“I see a lot of organisations here trying to foster that collaboration, which I really commend and encourage.”
It was a pleasure to hear these insights from Latifat, and we hope that you enjoy the interview.
For more conversations with our experts from the Congress in April do make sure you subscribe for weekly updates here!

by Charlotte Kilpatrick | Mar 27, 2024 | Infection |
Amid an “unprecedented multi-year upsurge” in global cholera cases, the International Coordinating Group (ICG) on Vaccine Provision is appealing for “immediate action” from the global community. This could include investments in access to safe water, sanitation, and hygiene, testing and detecting outbreaks quickly, improving quality of and access to healthcare, and fast-tracking additional production of the affordable oral cholera vaccine (OCV). The ICG is responsible for managing the global cholera vaccine stockpile, comprising the International Federation of Red Cross and Red Crescent Societies, Mèdecins Sans Frontières, UNICEF and WHO. The stockpile and delivery of vaccines is financed by Gavi.
A global increase
The current increase in cholera cases has been observed since 2021; 473,000 cases were reported to WHO in 2022, which is “more than double those reported in 2021”. Preliminary data for 2023 “reveal further increases”, with over 700,000 cases reported. Some of the outbreaks have high case fatality rates that exceed the 1% threshold used as an indicator for “early and adequate treatment” of patients with cholera.
“These trends are tragic given that cholera is a preventable and treatable disease and that cases had been declining in previous years.”
Countries that are currently the most affected include the Democratic Republic of the Congo, Ethiopia, Haiti, Somalia, Sudan, Syria, Zambia, and Zimbabwe.
Factors behind the increase
Cholera is an acute intestinal infection spread through food and water contaminated with faeces that contains the Vibrio cholerae bacterium. WHO states that the rise in cholera cases is “being driven by persistent gaps in access to safe water and sanitation”.
“Although efforts are being made to close these gaps in places, in many others the gaps are growing, driven by climate-related factors, economic insecurity, conflict, and population displacement.”
What can be done?
“Now more than ever, countries must adopt a multisectoral response to fight cholera.”
The ICG Members call on “currently and potentially affected countries” to take “urgent steps” to ensure that all populations have access to clean water, hygiene, and sanitation services, as well as the information required to prevent any cholera spread. This “requires political will and investment” at country level.
“This includes creating capacity for early detection and response, enhanced disease detection, rapid access to treatment and care, and working closely with communities, including on risk communication and community engagement.”
Pressure on the stockpile
Alongside water and sanitation requirements there is a “severe gap in the number of available vaccine doses” in comparison with the current need. Between 2021 and 2023 more doses were requested for outbreak response than had been needed for the “entire previous decade”. Indeed, in October 2022, the shortage necessitated the ICG to recommend a single vaccine dose rather than the “previous, long-standing two-dose regimen”.
Around 36 million doses were produced last year, but 14 affected countries registered a need for 72 million doses within a one-dose reactive strategy.
“These requests understate the true need.”
Preventative vaccination campaigns have been delayed to save doses for emergency outbreak control efforts. This creates a “vicious cycle”.
Global production capacity in 2024 is forecast to be 37-50 million doses but will “likely continue to be inadequate”. There is only one manufacturer that currently produces the vaccine: EuBioligics. Although the company is “doing its utmost to maximise output”, the ICG requires more doses. No new manufacturers are expected to join the market before 2025.
“The same urgency and innovation that we saw for COVID-19 must be applied to cholera.”
The ICG appeals to the vaccine and health community to “prioritise an urgent scale-up of vaccine production” and to “invest in all the efforts needed to prevent and control cholera”.
To participate in important discussions about how vaccines can be used to control outbreaks and prevent emergencies do join us at the Congress in Washington this April. Don’t forget to subscribe to our newsletters here!

by Charlotte Kilpatrick | Mar 25, 2024 | Global Health |
In an article for Journal of Evaluation in Clinical Practice, Dr David Robert Grimes and Professor Trisha Greenhalgh consider the argument that regulatory bodies have a “duty to act against” doctors who “intentionally mislead over the safety and efficacy of vaccination”. While enjoying a “uniquely privileged position of public trust” across much of the world, some doctors have “emerged as perpetrators” of both “inadvertent misinformation” and “deliberate disinformation” about vaccines, with “detrimental implications”. In their article, Dr Grimes and Professor Greenhalgh explore some notable examples of doctors speaking against the mainstream narrative.
Misinformation and the media
The authors reflect that “anti-vaccine rhetoric on social and mainstream media” has had a “substantial impact” on the uptake of COVID-19 vaccines, particularly in “certain demographic groups”, with social media having a key influence on “public perception of vaccine safety”. They recognise that the “abundance” of anti-vaccine materials allows “the weight of reputation” to give “undeserved legitimacy” to a “fiction”.
Trust the professionals
“The highly trusted nature of medical professionals means they are privileged to play a critical role in combatting these fictions and reversing vaccine hesitancy.”
While previous examples of concerted public engagement demonstrate the power of medical professionals, the COVID-19 pandemic “saw the worrying rise” of medical and scientific professionals “who made assertions” about vaccines based on “selective, questionable, or already refuted” evidence. As is often the case, these assertions were delivered in emotive language and repeatedly, to “huge audiences”. The authors therefore suggest that “how medical regulators should respond” is an “open, urgent question”.
“With medico-scientific professionals viewed as highly credible sources, their pronouncements on vaccination (both positive and negative) garner significantly more attention online than those of other individuals, and there is increasing awareness from both the public and medical bodies that false assertions from doctors do disproportionate harm to public understanding.”
A tragic example is offered in the paper: in the 1990s, Thabo Mbeki’s government “embraced AIDS denialism” in South Africa, citing “unorthodox opinions” of the scientist Peter Duesberg. This resulted in “an estimated 343,000 avoidable deaths”. Another example that continues to have consequences relates to vaccination: the “infamous” 1998 paper by Andrew Wakefield that “falsely linked” the MMR vaccine to autism. Although the paper was “exposed as fraudulent and retracted” in 2010, it has a “damaging legacy”. Furthermore, Wakefield was removed from the medical register by the GMC for research fraud, rather than the “long-term damage to public health” he caused through “deliberate spreading of vaccine disinformation”.
“The world is now a very different place. Social media platforms are now a major source of information and connection for millions of people around the world.”
During the “first ‘digital’ pandemic”, social platforms became outlets for “accurate, measured advice” but also presented opportunities to “garner huge audiences”.
Everything in moderation
While the authors emphasise the dangers that can be associated with allowing doctors to express their views widely, they recognise that “curtailing” their speech “risks suppressing clinical and scientific debate”. They present a handful of examples that demonstrate the importance of allowing doctors and scientists to challenge the norm. These include the case of Drs Graning and Dyar from the US, who “sounded the alarm” about a batch of polio vaccine in 1955.
However, the authors invite medical regulators to “draw the line between freedom of speech and protection of the public”. A key concern here is “whether statements appear to have been made in good faith”.
“Medical professionals, like all citizens, are entitled to their own opinion – but not their own facts.”
Authority comes not just from training and qualifications but the “reasonable assumption” that doctors will “accurately reflect what they believe to be the totality of evidence, conveying the spectrum of opinion and scientific uncertainty on contentious matters”.
The role of regulators
The authors suggest that medical regulators will need to adapt to the “challenges of health disinformation and maintaining standards”, particularly as “anti-vaccine views grow even inside the medical profession”. They consider the UK as a “no qualms” example in “dealing assertively” with complaints; in July 2023 a GP and homeopath who had encouraged parents to falsify children’s vaccination records and spread misinformation for over 15 years was “struck off” by the GMC. However, they highlight a “less proactive stance on members who make antivaccine statements in public”.
In the US, the situation is “also somewhat polarised”, with tensions between regulatory bodies who “take this issue very seriously” and the country’s “long tradition of supporting freedom of speech”. Indeed, a California law passed in 2022 that tackled misinformation from doctors was repealed a year later after doctors referred to their First Amendment rights.
“These conflicts highlight that regulation of what doctors say and write is a fine line to tread, particularly in the unregulated world of social media.”
Despite this, the authors demand “some regulatory safeguards” against those who “persistently ignore or distort data to present disingenuous positions to the public”.
What’s to be done?
The authors note that the UK GMC has “on occasion” implied that an argument against investigating high-profile doctors who make anti-vaccine statements is the potential to “amplify” their reach through publicity. However, they dismiss this as an effective argument as “celebrity doctor” already have “vast media reach for their fringe positions”.
“Regulatory action against doctors will not solve the complex problem of rising anti-vaccine sentiment among the lay public. But a rebuke from the regulator, should an investigation deem this appropriate, will send a clear signal to the public.”
The authors conclude that the current era demands regulatory involvement from an ethical and legal perspective. Furthermore, a public stand against the “propagation of misinformation” will support a regulatory body’s own credibility and “bolster public trust”.
Many members of our community publicly and privately engage in information dissemination and management to ensure that the public is presented with facts or an informed interpretation of these facts. However, this is a challenging task, so what can regulatory bodies do to encourage or support this? Do let us know your thoughts on this and the questions raised by the article and don’t forget that there’s still time to enter these discussions at the Congress in Washington this April. You can also subscribe to our newsletters here for more insights.

by Charlotte Kilpatrick | Mar 13, 2024 | Infection |
A statement from the World Organisation for Animal Health in March 2024 describes the “unprecedented milestone” reached by highly pathogenic avian influenza (HPAI), which is confirmed to have “traversed continents” to Antarctica’s mainland.
“The gravity of the situation intensifies as it infiltrates the northern tip of Antarctica’s mainland.”
This incidence was earlier reported by Argentinean scientists at the Primavera base and has been recorded on the World Animal Health Information System (WAHIS). A South Polar skua, a large seabird that breeds in sub-Antarctic and Antarctic zones, was found dead and tested positive. This is concerning as skuas are a potential vector as they migrate further north when not breeding.
A “new normal”
WOAH comments that while HPAI was previously considered “primarily a threat to poultry” it has “ushered in a ‘new normal’” as it now moves from wild birds to wild mammals with consequences “beyond anything previously seen”. WOAH’s most recent estimates suggest that 485 species from over 25 avian orders have been affected and 37 new mammal species infected since 2021. The Pacific Islands, Australia, and New Zealand are the only regions that WOAH considers “free of the disease”, recognising that the “situation is changing rapidly”.
Protection and prevention
Although HPAI outbreaks have resulted in “marked declines” in wildlife populations, WOAH identifies strategies for both long- and short-term protection of wildlife. For example, improved biosecurity, improved surveillance systems, and coordinated, interagency-multisecotral approaches can prevent spillover events and contain them when they do occur. WOAH recommends its own guidelines on the emergency vaccination of wild birds of high conservation importance against HPAI and the management of HPAI in marine mammals.
“The loss of wildlife at the current scale presents an unprecedented risk of wildlife population collapse, creating an ecological crisis.”
WOAH “encourages” Members to “quickly and thoroughly respond to outbreaks” and emphasises the importance of incorporating wildlife health into animal health surveillance, reporting, preparedness, and response systems.
“These efforts require not merely viewing wildlife as a potential risk to production animal and human health, but as beings warranting protection in their own right.”
Within the veterinary vaccines and One Health track at the Congress in Washington we look forward to hearing from Dr Daniel Peréz on the challenges and opportunities associated with mass vaccination against avian influenza. Get your tickets here to participate in these discussions, and don’t forget to subscribe to our newsletters for more insights.

by Charlotte Kilpatrick | Mar 13, 2024 | Global Health |
In March 2024 the United Nations Inter-agency Group for Child Mortality Estimation (UN IGME) shared a report that indicates that the number of children who died before their fifth birthday reached a “historic low” in 2022. The number dropped to 4.9 million, which has been described by health leaders as “progress”. However, there is evidently more to be done to ensure that more children survive early childhood and go on to live healthy and happy lives.
8 facts
The report begins with “8 things to know about newborn, child, adolescent, and youth mortality”.
- Good progress has led to fewer children dying before turning 5 than ever recorded.
- The annual death toll among children, adolescents, and youth remains unacceptably high.
- Children endure unequal chances of survival based on where they live, their socio-economic group, and if they live in a fragile and conflict-affected setting.
- Unless urgent action is taken to end preventable newborn and child deaths, many low- and lower-middle-income countries will not meet the Sustainable Development Goal (SDG) targets for newborn and under-five mortality and 35 million under age 5 will die before 2030.
- Progress is possible, even in low- and lower-middle-income countries.
- Investments at local, subnational, and national levels must be made to ensure proven interventions are available and accessible in every community, particularly where newborns and children are most at risk.
- Data are least available in the places where children face the highest risks.
- Global action is needed now to ensure progress – not complacency – defines the remaining years of the SDG era.
Data highlights from the report
- In 2022 neonatal deaths reached 2.3 million, which means one death every 14 seconds.
- In 2022 deaths of children under 5 reached 4.9 million, which means one death every 6 seconds.
- In 2022 adolescent deaths reached 0.9 million, which means one death every 35 seconds.
Comparing previous recorded years, the report tracks total deaths for earlier decades. From 1990-1999 there were 115 million deaths; from 2000-2009 there were 85 million deaths; from 2010-2019 there were 62 million deaths. Between 2020 and 2022 there have been 15 million deaths.
The risk of death for children under 5 was 18 times greater in sub-Saharan Africa than in Australia and New Zealand. The country with highest risk of mortality for children under 5 had 80 times greater risk than the country with the lowest risk.
- 4 out of 5 deaths in children under 5 occurred in sub-Saharan Africa and Southern Asia, but only 3 in 5 live births occurred in those regions.
- Sub-Saharan Africa accounted for 57% of global deaths under 5, but only accounted for 30% of global live births.
- Southern Asia accounted for a further 26% of global deaths under 5, but only accounted or 27% of global live births.
SDGs
The report states that, without “urgent action”, there is a risk of missing the SDG mortality targets of 25 or fewer under-5 deaths per 1,000 live births by 2030 (SDG 3.2.1) and 12 or fewer neonatal deaths per 1,000 live births by 2030 (SDG 3.2.2). At present, 59 countries are at risk of missing the SDG under-5 mortality target and 64 countries are at risk of missing the SDG neonatal mortality target.
- If we continue at the current rate, 35 million children under 5 are projected to die by 2030.
- Furthermore, 16 million children, adolescents, and youth between 5-24-years-old are projected to die by 2030.
- 9 million under-5 deaths could be prevented by accelerating progress to meet the SDG target in the countries at risk of missing the target.
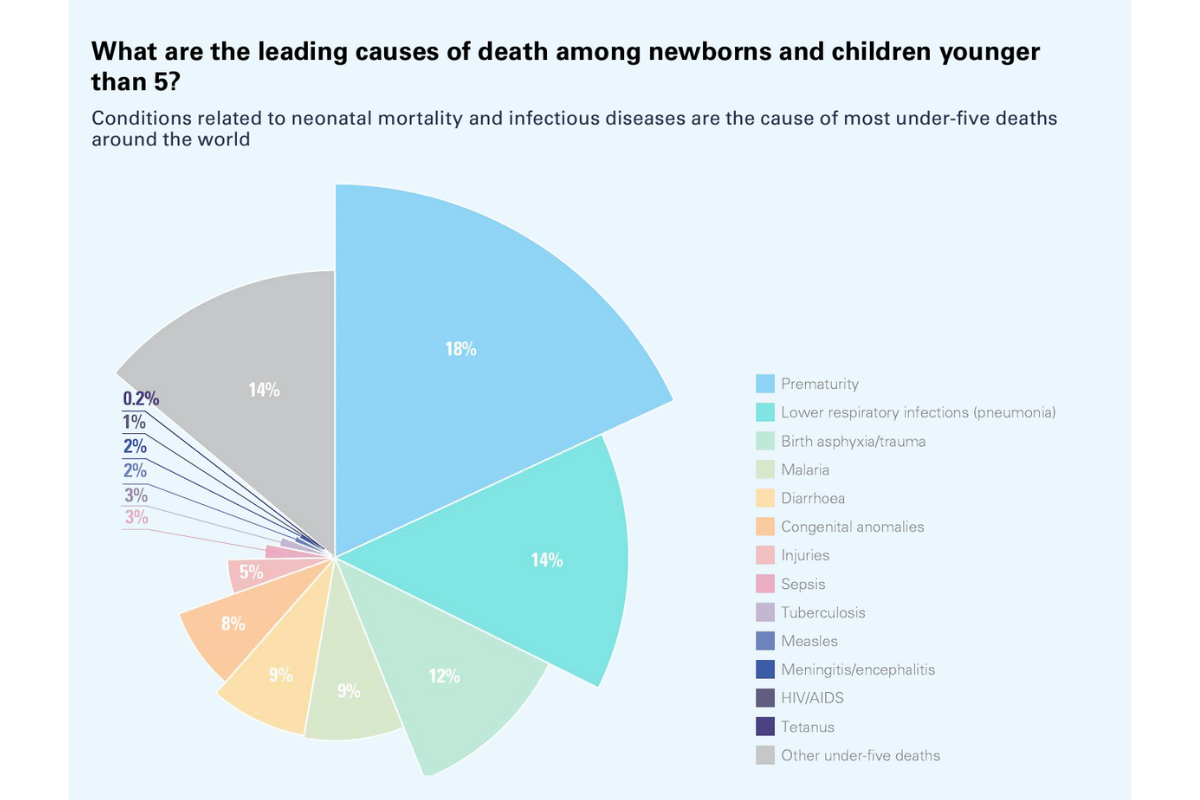
Experts weigh in
UNICEF Executive Director Catherine Russell recognised that “behind these numbers lie the stories of midwives and skilled health personnel” who help deliver newborns, alongside the health workers and community health workers who provide vaccination and health and nutrition support.
“Through decades of commitment by individuals, communities, and nations to reach children with low-cost, quality, and effective health services, we‘ve shown that we have the knowledge and tools to save lives.”
However, Dr Tedros Adhanom Ghebreyesus, WHO Director General, emphasised that despite “welcome progress” there are still “millions of families” who “suffer the devastating heartbreak of losing a child”.
“Where a child is born should not dictate whether they live or die. It is critical to improve access to quality health services for every woman and child, including during in emergencies and in remote areas.”
Dr Juan Pablo Uribe, Global Director for Health, Nutrition, and Population, World Bank, and Director, Global Financing Facility for Women, Children, and Adolescents, commented that the report is an “important milestone”.
“But this is simply not enough. We need to accelerate progress with more investments, collaboration, and focus to end preventable child deaths and honour our global commitment. We owe it to all children to ensure they have access to the same health care and opportunities, regardless of where they are born.”
UN Under-Secretary-General for Economic and Social Affairs agreed that the data are “important to track progress”, but also emphasise a need for more work.
“They should also remind us that further efforts and investments are needed to reduce inequities and end preventable deaths among newborns, children, and youth worldwide.”
The full report can be accessed on the UN IGME page here. How might vaccination strategies factor into efforts to reduce the number of deaths and meet SDGs? For more on this, join us at the Congress in Washington this April or subscribe to our newsletters here.