by Charlotte Kilpatrick | Oct 21, 2024 | Global Health |
In October 2024 WHO certified Egypt as malaria-free after a “nearly 100-year effort” by the government and people to end the disease. WHO described this as a “significant public health milestone” for the country’s more than 100 million inhabitants. Egypt is the third country to receive this certification in the WHO Eastern Mediterranean Region, following the United Arab Emirates and Morocco.
Across the globe, 44 countries and 1 territory have achieved this status by proving beyond reasonable doubt that the chain of indigenous malaria transmission by Anopheles mosquitoes has been interrupted nationwide for at least the previous three consecutive years. A country must also demonstrate its capacity to prevent the re-establishment of transmission.
Malaria becomes history
WHO states that malaria has been traced back as far as 4000 BCE in Egypt; there is genetic evidence of the disease in Tutankhamun and other ancient Egyptian mummies. More recently, efforts to reduce human-mosquito contact began in the 1920s with the prohibition of rice cultivation and agricultural crops near homes. With much of the population living along the banks of the Nile River and malaria prevalence “as high as 40%”, malaria was designated as a notifiable disease in 1930.
By 1942, malaria cases in Egypt exceeded 3 million due to population displacement caused by the Second World War, the disruption of medical supplies and services, and the invasion of Anopheles arabiensis, which is a “highly efficient mosquito vector”. Egypt responded to the outbreak by establishing 16 treatment divisions and recruiting more than 4000 health workers. The Aswan Dam, completed in 1969, brought an additional risk of malaria as standing water provides a mosquito breeding ground. Thus, Egypt worked with Sudan to launch a “rigorous” vector control and public health surveillance project.
By 2001, malaria was “firmly under control”, encouraging the Ministry of Health and Population to work on preventing the re-establishment of local malaria transmission. Egypt “rapidly” contained a small outbreak in the Aswan Governorate in 2014. The recent certification recognises continued efforts and initiatives including the free provision of malaria diagnosis and treatment to the population, regardless of legal status, and health professionals’ training to detect and screen for malaria. The country also has “strong” cross-border partnerships with neighbours like Sudan, which have been “instrumental”.
The beginning of a new phase
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, congratulated Egypt on its achievement.
“Malaria is as old as Egyptian civilisation itself, but the disease that plagued pharaohs now belongs to its history and not its future. This certification of Egypt as malaria-free is truly historic, and a testament to the commitment of the people and government of Egypt to rid themselves of this ancient scourge.”
Dr Tedros hopes that this will be an “inspiration to other countries in the region”, showing “what’s possible with the right resources and the right tools”. Deputy Prime Minister of Egypt H.E. Dr Khaled Abdel Ghaffar commented that the certification is “not the end of the journey but the beginning of a new phase”.
“We must now work tirelessly and vigilantly to sustain our achievement through maintaining the highest standard for surveillance, diagnosis and treatment, integrated vector management, and sustaining our effective and rapid response to imported cases. Our continued multisectoral efforts will be critical to preserving Egypt’s malaria-free status.”
Dr Abdel Ghaffar reaffirmed that the country will “continue with determination and strong will”. WHO Regional Director for the Eastern Mediterranean Dr Hanan Balkhy emphasised that the success is “not just a victory for public health but a sign of hope for the entire world”, including other endemic countries in the region.
“This achievement is the result of sustained, robust surveillance investments in a strong, integrated health system, where community engagement and partnerships have enabled progress. Furthermore, collaboration and support to endemic countries, such as Sudan, remain a priority.”
For insights into the latest malaria vaccine development progress, join us at the Congress in Barcelona next week. Don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 14, 2024 | Global Health |
Phase II trials of a “promising” human vaccine candidate against Rift Valley fever are beginning in Kenya with CEPI support. CEPI reported in October 2024 that this is the “most advanced stage of testing” for a human Rift Valley fever vaccine in an “outbreak-prone area”. Scientists at the University of Oxford and the Kenya Medical Research Institute (KEMRI)-Wellcome Trust Research Programme are leading the trial with $3.7 million funding from CEPI.
Rift Valley fever
First identified in Kenya’s Rift Valley, Rift Valley fever is usually found in people after direct contact with infected animals or bites from infected mosquitoes. Most infected people experience “mild disease”, but a small proportion develop the “severe haemorrhagic form”, with a risk of blindness, convulsions, encephalitis, and bleeding. In these cases, mortality rates can reach 50%.
Rift Valley fever has been detected across “much of Africa” and in the Middle East. It is mosquito-borne, which makes it “climate sensitive”. There is therefore a risk of outbreaks spreading to new areas or increasing in frequency or size. There are Rift Valley fever vaccines for animals, but no currently available or licensed vaccines for human use; the disease is a priority disease for R&D for WHO and CEPI.
ChAdOx1 RVF in trial
The vaccine, known as ChAdOx1 RVF, is based on the University of Oxford’s ChAdOx1 vaccine platform. It has shown positive results in healthy adults in the UK, meeting “many of the optimal product characteristics” of a WHO target product profile. It is one of three Rift Valley fever candidates in CEPI’s portfolio. 240 healthy adults will participate in the research, following local trial approvals.
Funding for the trial comes under CEPI’s strategic partnership with the University to accelerate the development of globally accessible vaccines against outbreak pathogens. Both organisations are “committed to enabling access to any vaccine outputs developed through this partnership”, including developing a target product profile suitable for low- and middle-income countries (LMICs), assessing the need for technology transfer, and priority supply to LMICs at an affordable price.
Professor George Warimwe, Principal Investigator of the trail and Deputy Executive Director of the KEMRI-Wellcome Trust Research programme, reflected that “nearly 100 years” after the disease was identified, there are “still no approved vaccines or treatments”.
“This vaccine trial brings us closer to addressing the rising frequency of outbreaks.”
Dr Richard Hatchett, CEO of CEPI, commented that Rift Valley fever “disproportionately affects the lives and livelihoods of vulnerable pastoral communities”, causing human fatalities and livestock losses.
“Investing in the promising human ChAdOx1 RVF vaccine diversifies CEPI’s portfolio and gives us a greater chance at protecting vulnerable populations against this worrisome threat that may become more prevalent with climate change.”
Director General of Africa CDC, H.E. Dr Jean Kaseya, agreed that the disease “leads to livestock losses and human fatalities, thus impoverishing communities who largely depend on livestock for their livelihood”.
“The launch of a Phase II clinical trial of a Rift Valley fever vaccine candidate in an endemic country is a crucial milestone in our efforts to control this disease. Africa CDC is proud to support this initiative that not only prioritises the health of our people but also demonstrates the continent’s growing leadership in advancing clinical research.”
Dr Kaseya stated that the ChAdOx1 RVF vaccine “offers hope to vulnerable populations” who are “disproportionately affected by the growing impact of climate change”.
Join us at the Congress in Barcelona later this month to explore efforts to address the growing challenges of climate change and infectious disease with vaccine development and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 3, 2024 | Global Health |
WHO announced in October 2024 that it is launching the Global Strategic Preparedness, Readiness, and Response Plan (SPRP) to tackle dengue and other Aedes-borne arboviruses. The Plan is intended to reduce the burden of disease, suffering, and deaths from dengue and other Aedes-borne arboviral diseases, like Zika and chikungunya, by “fostering a global coordinated response”. It presents priority actions to control transmission and offers recommendations to affected countries across various sectors. With five “key components”, the Plan is to be implemented over one year until September 2025, demanding US$ 55 million.
“The SPRP is a call to action for all stakeholders – from government agencies and health-care providers to communities and individuals – to join forces in the fight against dengue and other Aedes-borne arboviruses, through innovation, new technologies, and improved vector control strategies.”
Turning the tide
In the foreword by WHO Director-General Dr Tedros Adhanom Ghebreyesus we learn that dengue has “afflicted humanity for centuries, and possibly longer”; the first report of a clinically compatible case is recorded in a Chinese medical encyclopaedia in 992. From a much more contemporary perspective, dengue has spread “rapidly” in the past 20 years, enabled by “increased global travel and the effects of climate change”. Between 2000 and 2019, WHO documented a “tenfold surge” in reported cases, to 5.2 million. Since then, the surge has continued; over 12.3 million cases were reported by the end of August 2024.
The global prevalence and effects of arboviruses like dengue are a “significant threat to public health”, particularly in tropical areas where they are endemic. Addressing this threat demands a “concerted, strategic, and informed response”, which the Director-General hopes to achieve with the SPRP, a “comprehensive plan” to outline ways of controlling Aedes-borne arbovirus transmission in affected countries.
“Our multifaceted approach emphasises integrated surveillance, laboratory diagnosis, vector control, community engagement, clinical management, and research and development.”
This approach should reduce the burden of disease, save lives, and minimise the socioeconomic consequences of these diseases. Furthermore, the Plan includes measures for “safe programming” to ensure interventions are “secure and do not exacerbate the risk” for those who are already vulnerable to disease or those involved in responding to the crisis. Dr Tedros states that prevention and control is a “shared responsibility”.
“Together, we can turn the tide against this disease, protect vulnerable populations, and pave the way for a healthier future.”
Understanding the threat
Dengue is a challenge across all of WHO’s regions, endemic in more than 100 countries. Various factors, such as unplanned urbanisation and the effects of climate change, fuel the spread of dengue and other Aedes-borne arboviruses, such as Zika and chikungunya, putting more than four billion people at risk. The growing threat must be addressed with a “robust and dynamic strategy” that accounts for the current global epidemiological landscape. This is complicated by the “still developing” global surveillance system.
Transmission drivers like the effects of climate change and population growth can explain the increase of these infections in some areas, but they also point to the need for a multisectoral approach to prevent and respond to outbreaks.
The Plan
The Plan is intended to “reduce the burden of disease and deaths from dengue and other Aedes-borne arbovirus diseases in all affected WHO regions”. The strategic objective is “to accelerate progress in preventing and controlling dengue and other Aedes-borne arboviral disease outbreaks worldwide”, with the following specific objectives:
- Strengthen global multisectoral coordination and collaboration among stakeholders and partners in preparedness, response, and resilience to dengue and other Aedes-borne arbovirus diseases
- Enhance the capacity of Member States in early detection, reporting, confirmation, and response to outbreaks of dengue and other Aedes-borne arboviruses
- Strengthen the capacity of Member States to implement effective vaccination and integrated vector management strategies for mitigating the transmission of dengue and other Aedes-borne arboviruses
The SPRP combines strategic interventions tailored to local contexts and leverages inter-stakeholder synergies to “confront the challenges” posed by these diseases and move closer to controlling them. The following “interconnected pillars” are included in the multidisciplinary approach:
- Leadership, coordination, planning, monitoring, and prevention of sexual misconduct
- Risk communication and community engagement (RCCE) and infodemic management
- Surveillance, case investigation, and contact tracing
- Travel, trade, and points of entry surveillance and control
- Laboratory and diagnostics
- Integrated vector management and WASH & IPC
- Clinical management and therapeutics
- Operational support and logistics
- Essential health services and systems
- Vaccination
- Research, innovation, and evidence
The 5Cs
The SPRP aligns with WHO’s 2023 Framework for Health Emergency Prevention, Preparedness, Response, and Resilience (HEPR) with a focus on five “core health emergency components”:
- Collaborative surveillance
- Strong national integrated disease, threat, and vulnerability surveillance,
- Effective diagnostics and laboratory capacity for pathogen and genomic surveillance
- Collaborative approaches for event detection, risk assessment, and response monitoring
- Community protection
- Community engagement, risk communication, and infodemic management
- Population and environmental public health interventions
- Multisectoral action for social and economic protection
- Access to countermeasures
- Fast tracked research and development
- Scalable manufacturing platforms
- Coordinated supply chains and emergency
- Emergency coordination
- Strengthened workforce capacity for health emergencies
- Strengthening health emergency preparedness, readiness, and resilience
- Health emergency alert and response coordination
- Safe and scalable care
- Scalable clinical care during emergencies
- Protection of health workers and patients
- Maintenance of essential health services
How do you think the SPRP can be effectively translated into specific contexts and implemented sustainably?
For insights into vaccination efforts for diseases that are being exacerbated by the effects of climate change, get your tickets to the Congress in Barcelona this month, or subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 19, 2024 | Infection |
The Times of India reported in September 2024 that cases of chikungunya in children were symptomatic of a “shift” in epidemiology. Case numbers have increased and are more frequently requiring hospitalisation, particularly among children. While these changes are cause for concern for some infectious disease experts, they are careful to emphasise the need for thorough research. Indeed, the increase in cases may be attributed to improved surveillance efforts.
Cases in children
The latest report suggests that in Maharashtra, a populous state in the west of India, cases have exceeded 2,600 since the start of the year. This is a 50% increase on the previous year, and the disease is “increasingly” affecting children, some of whom require time in intensive care. Dr Sachin Shah, neonatologist at Surya Hospital in Pune, is concerned by the number of children suffering:
“In my OPD for older children, it is the most common complaint this week.”
Five newborns were diagnosed with chikungunya in Pune, two of whom acquired it from their mothers. Dr Shah described high-grade fever, elevated liver function, and low appetite among the symptoms. One neonate developed a “life-threatening disorder” characterised by hyper-inflammation. Infectious disease specialist at H N Reliance Hospital in Girgaum notes the change in severity among patients who are admitted to hospital.
“Chikungunya has acquired a notorious edge this year.”
This change is also observed by Dr Anita Mathew, infectious disease specialist at Fortis Hospital, Mulund, who has had patients require ICU attention.
“The extreme symptoms are new to Mumbai, but are not unknown in medical literature.”
Is this new?
While some experts are questioning this “shift” in disease, others are wary of sounding the alarm. Dr Radhakrishna Pawar of the state health department acknowledged the change in clinical appearance but urged further investigation.
“We need to study it in detail before making such claims.”
Furthermore, while cases have increased, Dr Pawar noted that they do not reach the numbers required to qualify as an outbreak or epidemic.
Vaccine access
Although there is a licensed chikungunya vaccine to address the “significant unmet medical need” presented by chikungunya, Valenva’s IXCHIQ is only currently approved in the United States, Europe, and Canada. However, Valneva is “focussed on expanding the vaccine’s label and access”, working with CEPI to support access in LMICs, post-marketing trials, and potential label extensions.
For the latest on infectious disease management with vaccine innovation, join us at the Congress in Barcelona next month, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Jun 3, 2024 | Infection |
In May 2024 the WHO shared a disease outbreak news update on the global dengue situation, stating that by 30th April 2024 over 7.6 million dengue cases have been reported to WHO in 2024. This total includes 3.4 million confirmed cases, over 16,000 severe cases, and over 3,000 deaths. The “substantial increase” in cases has been “particularly pronounced” in the Region of the Americas, where the number of cases exceeded 7 million by the end of April 2024. This surpasses the annual high of 4.6 million cases in 2023. 90 countries have known active dengue transmission this year.
However, many endemic countries do not have strong detection and reporting mechanisms, which means that the true global burden of dengue is underestimated. WHO states that “real-time robust dengue surveillance” is needed to control transmission “more effectively”. This would address concerns about undetected cases, co-circulation and misdiagnosis as other arboviruses, and unrecorded travel movements.
“The overall capacity for countries to respond to multiple, concurrent outbreaks continues to be strained due to the global lack of resources, including shortages of good quality dengue diagnostic kits for early disease detection, lack of trained clinical and vector control staff, and community awareness.”
WHO maintains that the overall risk at the global level is “high”, with dengue remaining a global threat to public health.
Co-circulation concerns
There is “considerable overlap” in the geographic distribution of dengue, chikungunya, and Zika viruses; these are all transmitted by Aedes mosquitoes and share some clinical features. This can result in misdiagnoses and misreporting in the absence of differential laboratory testing. Surveillance systems that specifically target transmission of chikungunya or Zika are “weak or non-existent” in many countries.
“As dengue, chikungunya, and Zika viruses share the same Aedes mosquito vectors and co-circulate in the same geographic areas, they also share many prevention strategies, such as differential diagnosis, mosquito control, and public awareness campaigns.”
However, WHO notes “important differences” between these diseases that affect risk populations, patient management, and use of health care resources. Therefore, expanding surveillance for all three viruses will help public health authorities determine the true burden of each more accurately and respond appropriately.
Risk assessment and advice
In November 2023 WHO assessed the global risk of dengue as high. In December, the internal emergency response was assigned as G3 at the global level.
“Given the current scale of the dengue outbreaks, the potential risk of further international spread and the complexity of factors impacting transmission, the overall risk at the global level is still assessed as high and thus dengue remains a global threat to public health.”
Vector control interventions are “key” to dengue prevention and control, and WHO states that vector control activities should target “all areas” with risk of human-vector contact, including residences, workplaces, schools, and hospitals. WHO promotes Integrated Vector Management (IVM) to control Aedes species. Personal protective measures during outdoor activities are also encouraged. While there is no specific treatment for dengue infection, WHO highlights that early detection and access to appropriate healthcare for case management reduces mortality.
Vaccination should be considered within an integrated strategy to control the disease; WHO recommends the use of TAK-003 in children aged 6-16 years in settings with high dengue transmission intensity.
For the latest on vaccination strategies against infectious disease do join us at the Congress in Barcelona or subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Mar 21, 2024 | Infection |
In March 2024 the University of Oxford announced a partnership with the University of Edinburgh to examine the immune system’s response to “repeated” malaria infections. The BIO-004 study is a collaboration between the Department of Biochemistry and Oxford Vaccine Group from Oxford and the University of Edinburgh’s Institute of Immunology and Infection Research and will offer a “unique insight” into how the immune system “adapts” after a few malaria infections to tolerate the parasites and develop natural immunity to severe illness.
BIO-004
The trial, hosted by the Oxford Vaccine Group, will infect adult volunteers who have never had malaria three times in “carefully monitored conditions”. Serial “malaria challenges” will be conducted over a 20-month period, allowing researchers to compare the immune response to all three infections. Although over 600 people have previously participated in malaria challenge studies in Oxford, this is the “word first” use of repeated malaria challenges to study the human immune system in “such close detail”.
Dr Angela Minassian is Chief Investigator for the trial and commented that malaria is a disease “caused by a parasite infection that is spread by mosquitoes”. While it causes “hundreds of thousands of deaths every year”, Dr Minassian reflected that 75% of these occur in children under the age of 5 years.
“Current efforts to control the disease are focused on stopping mosquitoes from biting, either by spraying insecticides or sleeping under bed nets, and by reducing the number of parasites in the blood, using drugs or the recently licensed malaria vaccines, RTS,S and R21. However, these measures are only partially effective.”
Dr Minassian suggests that the first malaria infection in life is “the most dangerous” with the immune system learning to adapt to the parasite over repeat infections. However, the mechanism by which this occurs is “unknown”.
“BIO-004 is an experimental medicine study funded by the UKRI-MRI which aims to answer this question and help inform strategies to protect young children in endemic countries from severe disease and death due to malaria.”
Principal Scientific Investigator, Edinburgh’s Dr Phil Spence, commented that “children who survive their first malaria infection quickly develop immunity” against the most severe forms of disease, which often prevents severe infection in the future.
“We now understand that, during the first malaria infection, the immune system launches a full-scale attack which causes collateral damage to healthy tissues. Then, with subsequent infections, the immune system learns to rein in this damaging response and tolerate the presence of parasites in the bloodstream.”
Dr Spence hopes that the trial will “change” the fact that doctors “do not currently know the mechanisms that allow the immune system to do this”.
“Understanding how ‘tolerance’ to disease is induced and maintained in malaria will, for the first time, allow us to harness the power of this defence strategy to design new policies, medicines, or vaccines that combat severe malaria in children and ultimately save lives.”
We’re looking forward to hearing more from our experts on malaria elimination at the Congress in Washington this April, so do join us if this is of interest and don’t forget to subscribe to our newsletters here.

by Charlotte Kilpatrick | Mar 21, 2024 | Infection |
In March 2024 the WHO African Region shared a “disease outbreak news” update on yellow fever (YF) in the region from the beginning of 2023. 13 countries have documented “probable and confirmed cases”, and although the overall risk at regional level has been reassessed as “moderate” and the global risk “remains low”, WHO calls for “active surveillance” due to the potential for onward transmission. The “urban proliferation of Aedes spp. mosquitoes can “significantly amplify transmission risks”, which would lead to “swift outbreaks”. However, thanks to the WHO-led global Eliminate Yellow Fever Epidemics (EYE) secretariat’s coordinated preventative and reactive efforts, vaccination coverage has been “substantially enhanced”. Around 62 million people have been vaccinated in Africa through mass vaccination campaigns.
Yellow fever
WHO describes yellow fever as an “epidemic-prone, vaccine-preventable disease”, which is caused by an arbovirus transmitted mainly through the bites of infected mosquitoes. The incubation period ranges from 3 to 6 days, but many people do not experience symptoms. For those who do, fever, muscle pain, and nausea are common.
A small percentage of cases progress to a “toxic phase” with systemic infection affecting the liver and kidneys. In these cases, patients can have more severe symptoms, such as high-grade fever, abdominal pain and vomiting, and jaundice and dark urine caused by acute liver and kidney failure. Bleeding can occur from the mouth, nose, eyes, or stomach. In half of cases with severe symptoms death can occur within 7 to 10 days.
Yellow fever can be prevented with an effective vaccine, which is described by WHO as “safe and affordable”; a single dose offers sustained immunity and life-long protection, without requiring a booster dose.
Cases in 13 countries
WHO reports that, from the beginning of 2023 until 25th February 2024, a total of 13 countries in the region have documented “probably and confirmed cases” of YF: Burkina Faso, Cameroon, the Central African Republic, Chad, Republic of the Congo, Côte d’Ivoire, the Democratic Republic of the Congo, Guinea, Niger, Nigeria, South Sudan, Togo, and Uganda. Preliminary data for 2023 indicate a case fatality rate (CFR) of 11% and a sex ratio of 1:7 (M: F). The median case age is 25.
“Suboptimal surveillance capacity exists in many affected countries, with data fragmentation, limited integration with routine surveillance and clinical systems, and a lack of standardised case definitions that contribute to underreporting and increased mortality rates.”
WHO’s risk assessment
Within efforts to monitor and respond to infectious disease outbreaks WHO conducted a Rapid Risk Assessment for yellow fever on 12th February 2024. This was intended to reassess the current regional risk of multiple ongoing YF outbreaks and provide recommendations for a more effective and coordinated response.
The regional level risk is described as “moderate” and the global risk “remains low”. However, WHO emphasises the need for “active surveillance” in the context of the potential for onward transmission through viraemic travellers and the presence of the competent vector in neighbouring regions.
For more discussions on infectious disease management, particularly encouraging greater uptake of vaccines for vaccine-preventable diseases, why not join us at the Congress in Washington this April, or subscribe to our newsletters here?

by Charlotte Kilpatrick | Mar 19, 2024 | Global Health |
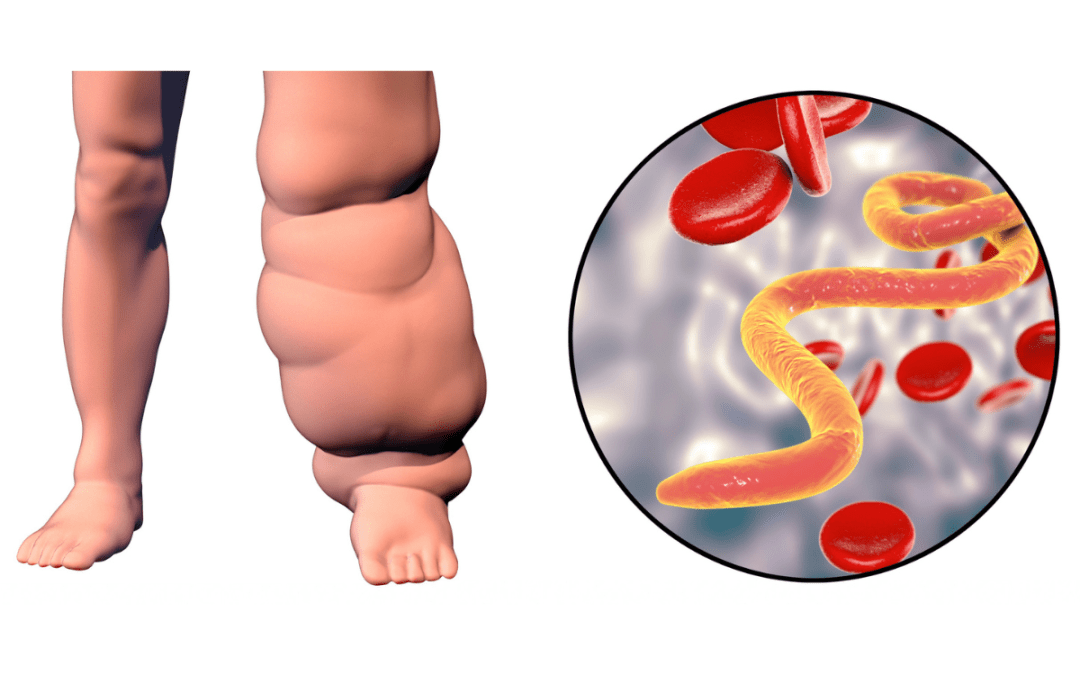
In March 2024 the University of Illinois Chicago announced that an NIH grant of $2.4 million will support UIC researchers as they bring a vaccine against elephantiasis to clinical trial. Professor Ramaswamy Kalyanasundaram, head of the UIC Medicine Rockford Department of Biomedical Sciences, is working towards the development of the first vaccine for lymphatic filariasis, which affects “more than 100 million people” worldwide. The grant is part of the NIH Small Business Innovation Research programme and will contribute to final studies required for investigative new drug approval from the FDA.
Elephantiasis
WHO describes lymphatic filariasis, also known as elephantiasis, as a “painful and profoundly disfiguring disease” caused by infection with nematode parasites in the Filariodidea family, transmitted through the bites of infected mosquitos. Mosquito-transmitted larvae are left on the skin, from where they migrate to the lymphatic vessels to develop into adult worms and continue a cycle of transmission.
In communities where filariasis is transmitted, “all ages are affected”, and it has a “major social and economic impact”. Lymphatic filariasis affects “over 120 million people” in 72 countries across the tropics and sub-tropics of Asia, Africa, the Western Pacific, and parts of the Caribbean and South America.
The CDC describes the disease as a “leading cause of permanent disability”, commenting that communities “frequently shun and reject” people who have been disfigured. Affected people are unable to work because of the disability, which “harms their families and their communities”.
A desperate need
Professor Kalyanasundaram emphasises that a vaccine is urgently needed, as medications for lymphatic filariasis are proving ineffective. Despite efforts to provide medication to areas where the disease is endemic, infections persist in many regions. In 2015, he spent five months in India to evaluate these efforts, finding that many people continue to suffer from the disease.
“There’s medicine available, but the medicine does not completely cure the infection and not everyone has access to or will take the drugs.”
Thus, a single shot vaccine that offers at least a year of immunity, is a promising alternative.
“If we have a vaccine, wherever you suspect that not everybody is taking the medication, you can at least give a shot and then they will be protected for at least a year.”
A long journey nears fruition
Professor Kalyanasundaram began working towards this goal in the 2000s as part of the WHO Genome Project and identified the parasite genes that “manipulate” the host’s immune system to evade a response and survive in the human body. His research group then identified vaccine antigens by finding which antibodies in the blood of naturally immune people protected against the genome.
“I’ve had several postdoctoral research associates and graduate students work on the project over the past 20 years. All of our work is finally coming to fruition.”
The team was able to identify potential targets and have tested these in preclinical studies for two decades. A previous NIH grant enabled PAI Life Sciences in Seattle to help with scale up of production of the current vaccine formula: a “combination of three antigens to induce a prolonged immune response that wards off infection”. This grant will fund toxicology studies and other final steps.
“We’re hoping that in the next three to five years, the vaccine should be ready for actual human immunisation.”
Neglected diseases such as elephantiasis will be considered during our keynote panel on priority pathogens and the inclusion of neglected diseases in a global strategy to tackle them; you can get your tickets to join us for this here, and don’t forget to subscribe here.