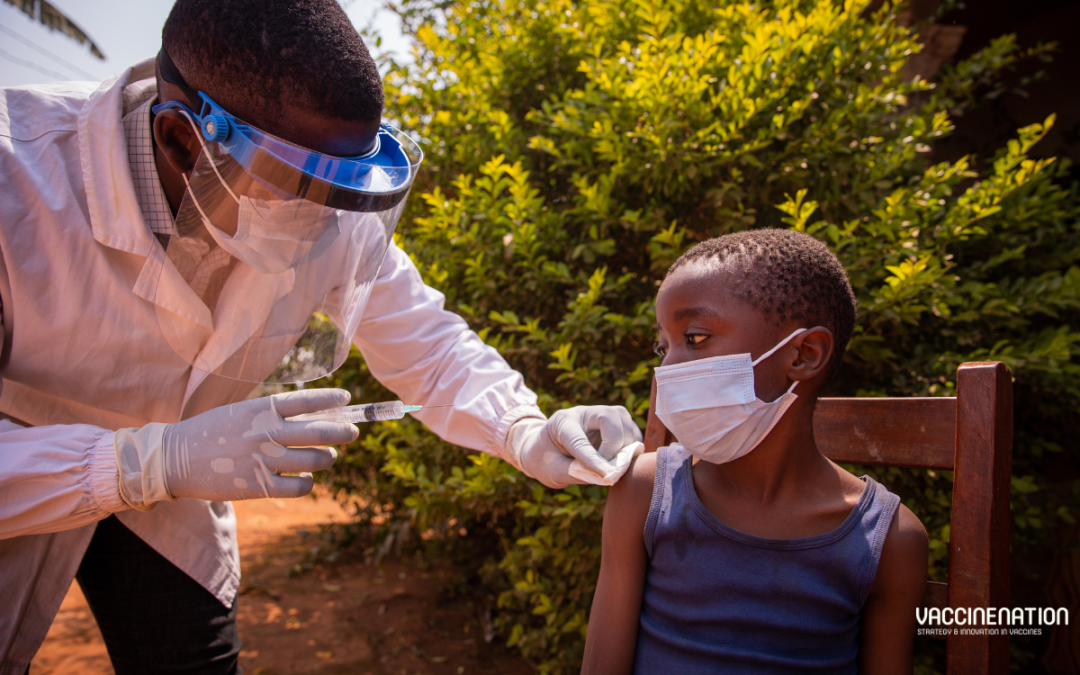
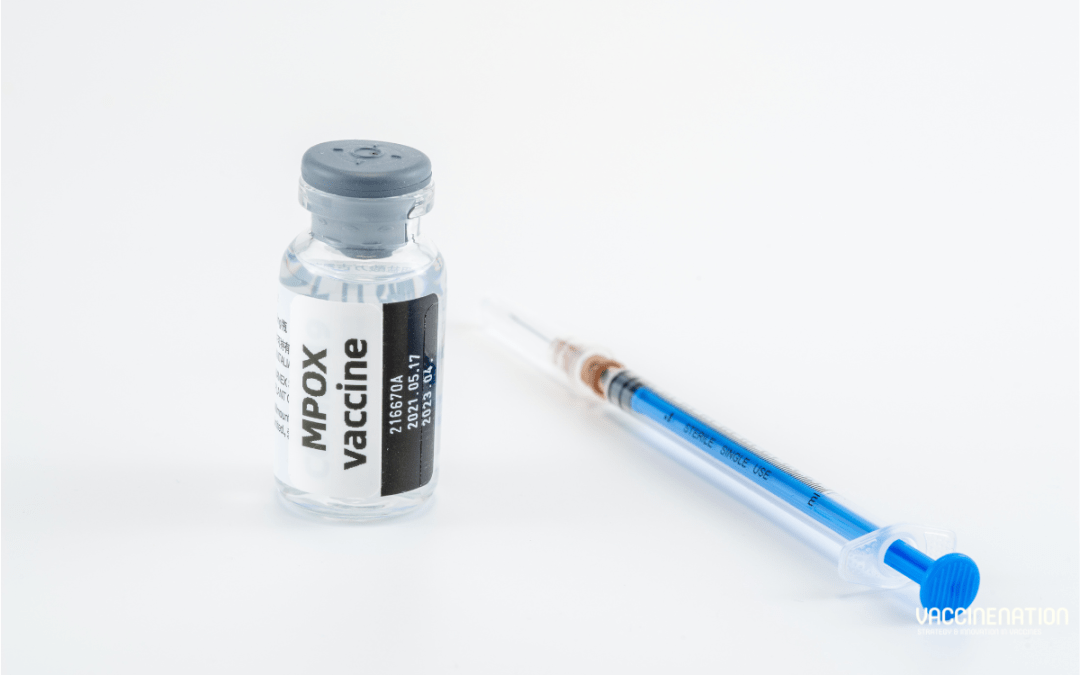by Charlotte Kilpatrick | Oct 23, 2024 | Global Health |
A study in The Lancet Global Health sought to provide counterfactual scenarios to evaluate the short-term effects of different vaccination strategies on mpox cases and deaths in the Democratic Republic of the Congo (DRC). The researchers used a dynamic transmission model to simulate mpox transmission, stratified by age and province; this was used to assess potential vaccination strategies and their effects on deaths and cases in an epidemic year. The results indicate that vaccinating children aged 15 years or younger, or younger than 5 years, in endemic regions, would be the “most efficient use of vaccines” when resources are limited.
Mpox in DRC
Mpox was first identified in the Democratic Republic of the Congo (DRC) in 1970; it is a zoonotic infectious disease caused by the monkeypox virus (MPXV), which is endemic in “numerous regions” of west and central Africa. MPXV has two clades:
- Clade I is endemic in central Africa with an estimated case fatality rate of up to 10% and mainly affecting children. It is divided into two subclades, Ia and Ib.
- Clade II was historically found in west Africa, with an estimated case fatality rate of up to 1%-3%. It is also divided into two subclades, IIa and IIb. Clade IIb was responsible for the global mpox epidemic in 2022.
The authors state that, until 2022, MPXV was not associated with large outbreaks; most cases were related directly to sylvatic transmission from animals to humans via hunting, wild game preparation, and consumption. Increases in human-to-human transmission were identified in 2017.
The researchers suggest that the low likelihood of transmission in the early decades after the virus’ discovery could be related to smallpox eradication programmes, which offered cross-immunity via vaccination against a related orthopoxvirus. Indeed, since the cessation of the smallpox vaccination programme in the DRC, there has been a “concurrent increase in mpox cases and outbreak frequency”. There is an ongoing, “unprecedentedly large” outbreak of clade I mpox in the DRC, with more than 14,000 reported suspected cases by the end of 2023 and a 4.6% case fatality rate. Over 70% of the deaths are in children younger than 15 years.
Genetic analyses of clade Ia MPXV genomes indicate that in this outbreak, multiple, independent zoonotic introductions into the human population have occurred from one or more reservoir species. An increasing burden of clade Ib MPXV infections have been identified in eastern DRC with evidence of “sustained” human-to-human transmission and many cases in women aged 15-29 years, but clade Ia infections continue to comprise most mpox cases in the DRC.
The study
Bavarian Nordic’s modified vaccinia Ankara vaccine (JYNNEOS) is protective against mpox. It was approved by the US FDA in 2019 but was not widely used against mpox until the 2022 outbreak, when it was “quickly mobilised to vaccinate people at high risk of infection in the USA and Europe”. Despite its high efficacy at two doses, it is “largely unavailable” outside the USA and Europe.
The authors aimed to inform policy and decision makers on the “potential benefits of, and resources needed,” for mpox vaccination campaigns in the DRC. They used an approach based on models from operations research and decision science to offer a robust analysis of policy choices “even in the context of incomplete and uncertain data”. The study uses mathematical modelling to simulate the spread of mpox in the DRC during 2023.
Without vaccination, the model predicted 14,700 cases of mpox and 700 deaths from mpox in the DRC over 365 days, consistent with reported estimates. Almost 50% of the cases and deaths came from the province of Equateur. Cases were evenly split between the three age groups: 34% in children under 5 years, 32% in children aged 5-15 years, and 34% in people older than 15 years. However, deaths were “predominantly” seen in children younger than 5 years (51%).
Vaccinating 80% of children younger than 5 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 10,500 cases and 400 deaths. Vaccinating in endemic provinces increased cases to 10,700 and deaths remained the same. The numbers of vaccine doses needed for the strategies were 41.4 million (all provinces), 33.8 million (provinces with a history of mpox), and 13.2 million (endemic provinces only).
Vaccinating 80% of children younger than 15 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 6,400 cases and 200 deaths. Vaccinating in endemic provinces increased cases to 6,800 and deaths remained the same. The numbers of vaccine doses required for these strategies were 81.6 million (all provinces), 67.1 million (provinces with a history of mpox), and 26.6 million (endemic provinces only).
Vaccinating 80% of all ages in all provinces or only non-endemic provinces with a history of cases decreased the case burden to 1,400 cases and 100 deaths, and 2,000 cases and 100 deaths when vaccinating in provinces endemic for mpox. The numbers of doses required for these strategies were 170.8 million (all provinces), 142.0 million (provinces with a history of mpox), and 56.8 million (endemic provinces only).
Managing resources
The paper finds that vaccinating all ages leads to the “largest impact on magnitude of cases and deaths”, but that vaccinating only children aged 15 years or younger provides “nearly the same effect with fewer vaccine doses required”. Although vaccinating only children younger than 5 years showed a “drop-off” in averted cases and deaths, it provides the most efficiency.
“This analysis shows the effectiveness of focussing an mpox vaccination campaign specifically in the provinces endemic for mpox in the DRC. This targeted strategy prevents nearly as many cases and deaths as broader approaches but uses fewer vaccine doses and thus would be less costly to implement.”
Alexandra Savinkina, fourth year PhD student in the Yale School of Public Health (YSPH) Department of Epidemiology (Microbial Diseases), commented that this study could influence vaccination policy.
“My hope is that it could help inform policy for vaccination in the country and potentially the region and move the needle forward on getting vaccines to the people who need them most in the DRC.”
Savinkina hopes that “we can learn from the global mpox outbreak that we can’t ignore disease in other places”.
“If the resources to help people exist, I think we should be using them, whether in the U.S. or in Africa.”
Dr Gregg Gonsalves, associate professor of epidemiology at YSPH, acknowledged barriers to access.
“We take it for granted that we can get a vaccination for COVID or a flu shot at our local CVS, but the infrastructure to deliver vaccines in DRC is far less robust.”
For more vaccine research updates, subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 8, 2024 | Infection |
A recent WHO situation report on the mpox PHEIC reveals that 14 countries on the continent have reported mpox cases in the last six weeks and are considered to have “active” outbreaks. The most affected country in 2024 is still the Democratic Republic of the Congo (DRC), which recently announced the start of an mpox vaccination campaign. The update presents reported confirmed mpox cases and deaths as well as reported suspected mpox cases, as defined by the countries that have reported them.
Risk levels
The report presents the mpox risk of geographical spread and potential impact on health in various regions:
- Eastern Democratic Republic of the Congo and neighbouring countries: high
- Areas of the Democratic Republic of the Congo where mpox is endemic: high
- Nigeria and other areas of West, Central, and East Africa where mpox is endemic: moderate
- All other countries in Africa and around the world: moderate
However, the report notes that individual country or regional bloc assessments may vary, and the risk could be assessed as low. Individual-level risk is “largely dependent on individual factors” like exposure risk and immune status.
Cases
By 29th September, 14 countries on the continent have reported mpox cases in the last six weeks. The most affected country in 2024 is the Democratic Republic of the Congo (DRC) with 5,610 confirmed cases and 25 deaths, followed by Burundi, with 853 confirmed cases and no deaths, and Nigeria, with 78 confirmed cases and no deaths. Although the epidemic curve of confirmed cases by country suggests a decline in reported cases in DRC, this trend “should be interpreted with caution” amid “reports of limited testing and stockout of testing supplies”.
Focus on North Kivu
As of 28th September 2024, the province of North Kivu in DRC had reported 323 confirmed mpox cases, including two confirmed deaths. After initial detection in May 2024 genomic sequencing analysis revealed clade Ib monkeypox virus (MPXV). There are 34 health zones in North Kivu; 14 have reported confirmed mpox cases, The most affected zones are Goma, Karisimbi, and Nyiragongo. 1,108 suspected mpox cases in North Kivu have been tested with a test positivity of 29%. The number of confirmed cases in the province continues to increase.
Among confirmed cases, 117 (36%) are individuals living in camps for Internally Displaced People (IDP), in the three most affected health zones. 13 IDP camps have reported at least one mpox case; the most affected are Munigi, Mugunga, Rusayo, and Kanyaruchinya. 8 out of 14 IDP camps (57%) have reported only one case, which indicates sporadic introduction, likely from outside the camp. Transmission may be sustained in the other six, which show more cases over time.
In North Kivu, around 50% of confirmed mpox cases are among adults and 54% of total cases are male. However, in IDP camps, approximately 75% of mpox cases are among children up to 17 years old and are evenly distributed between males and females. WHO believes that mpox transmission in North Kivu is “exclusively” human-to-human, mainly at the community level. 117 out of 323 (36%) cases have a known epidemiological link. The mode of transmission for 109 cases (34%) is reported to be sexual contact; among these cases, 57 (52%) are reported among female sex workers.
216 out of 323 (67%) cases have recovered from the disease; 109 are in isolation, 33 in the household, and 76 in healthcare facilities. 19 (9%) cases have presented complications and two have died in hospital.
Clade Ib detected in India
On 1st October 2024, the Ministry of Health and Family Welfare of India notified WHO of the first mpox case due to MPXV clade Ib. The National Focal Point reports that the case is an adult male, Indian national, with a recent history of international travel to the United Arab Emirates (UAE). The patient developed mpox symptoms on 8th September in UAE before arriving in India on 13th September. On 16th September he was admitted to a public hospital.
On 19th September, samples were tested at the National Institute of Virology (NIV) in Pune, confirming MPXV infection. The patient recovered without complications and was discharged on 30th September 2024. The health authorities in UAE are conducting a detailed case and contacts investigation to finalise the “comprehensive verification and validation process as per IHR procedures”.
This is the second reported case of this clade MPXV infection in the WHO South-East Asia Region (SEAR) and the third reported case of clade Ib MPXV infection outside the African Region. However, the first clade Ib infection in SEAR, reported in Thailand, and the clade Ib infection in Sweden, had a recent history of travel to affected countries in Africa, which this case did not.
Vaccine updates
WHO indicates that it is finalising the issuance of prequalification age extension of MVA-BN for persons 12-17 years old after European Medicines Agency (EMA) authorisation. It is providing “strategic and technical support” to the African Vaccine Regulatory Forum (AVAREF) and issuance of emergency use authorisation for MVA-BN to national regulatory authorities. In collaboration with AFRO, DRC, Ghana, Nigeria, Rwanda, and Tanzania, WHO is harmonising the cohort safety event monitoring protocol following mpox vaccination and ensuring global data collection.
With receipt of 265,000 doses of MVA-BN, DRC has begun a vaccination campaign in North Kivu with the intention of expanding to 11 of the most affected health zones across various provinces.
Join us at the Congress in Washington in April next year to reflect on the global response to this outbreak and hear updates on continued mpox vaccine development, and don’t forget to subscribe to our weekly newsletters for more insights.

by Charlotte Kilpatrick | Oct 7, 2024 | Global Health |
WHO Africa reported in October 2024 that the Democratic Republic of the Congo (DRC) has started a vaccination campaign as part of outbreak control efforts against mpox. The vaccination drive has launched in the eastern North Kivu province and will prioritise health workers and frontline responders, contacts of confirmed cases, contacts of those contacts, and other at-risk groups. It will later be implemented in 11 of the most affected health zones in Equateur, North Kivu, Sankiri, South Kivu, Sud-Ubangi, and Tshopo provinces.
Addressing the emergency
The Democratic Republic of the Congo (DRC) has reported more than 30,000 suspected and laboratory-confirmed cases and 990 deaths since the start of 2024. These numbers account for 90% of the cases reported from 15 countries in the African region this year. WHO recommends that vaccination should form part of a “comprehensive response” involving enhanced surveillance, community engagement, and case management. It is working with partners and the national authorities to “scale up and reinforce all the key control measures”.
In preparation for the mpox vaccination campaign, WHO has supported national health authorities in training health workers, enhancing vaccine delivery systems and infrastructure, and community engagement. There are also efforts to “reinforce measures to identify and address” vaccine misinformation and disinformation, responding with increased access to accurate information.
Delivering doses to affected areas
Noting that mpox vaccines are “currently in short supply, especially in Africa”, WHO is encouraging global collaboration to get doses to the people who need them most. In September, WHO prequalified MVA-BN for mpox, which is “expected to facilitate timely and increased access”. It is also working with partners like Gavi and UNICEF to establish a distribution mechanism for donated doses and direct procurements. DRC has received 265,000 doses of MVA-BN, donated by the European Commission’s Health Emergency Preparedness and Response Authority, Gavi, and the United States Government.
WHO Regional Director for Africa, Dr Matshidiso Moeti, expressed gratitude to these partners for their donations.
“As we rally efforts to stop the mpox outbreak, the rollout of the vaccine marks an important step in limiting the spread of the virus and ensuring the safety of families and communities.”
Dr Moeti commented that WHO is “working closely with the national authorities to effectively deliver the vaccines to those who need them most”. Africa CDC also recognised the collaborative effort, which “underscores the collective global commitment” to controlling the outbreak in Africa. H.E. Dr Jean Kaseya, Africa CDC Director General, commended the DRC’s “swift action” in launching the campaign, which “showcases the strength of its public health leadership”.
“By prioritising vulnerable populations, including frontline health workers and those most at risk, the country is taking critical steps to contain the outbreak. Africa CDC remains committed to working closely with the DRC to ensure vaccines reach those who need them the most, while also working to strengthen health systems to prevent future outbreaks. Our top priority is to secure safe and effective vaccines for children in the next phase of vaccination.”
Mpox vaccination will be a key area of high-level discussions at the Congress in Washington next April, including on a keynote panel that will consider the “role of vaccines in a changing world”. Get your tickets to join us for these conversations, and don’t forget to subscribe to weekly vaccine updates here.

by Charlotte Kilpatrick | Sep 30, 2024 | Infection |
In September 2024, WHO’s Regional Office for Africa announced that Rwandan health authorities are “intensifying outbreak control efforts” after detecting Marburg virus disease in the country for the first time. 26 cases have been confirmed in seven of the country’s districts, and six deaths have been reported. The health authorities are implementing “comprehensive response measures” and an investigation to determine the origin of the outbreak. WHO states that it is supporting these efforts with expertise and tools.
Cases reported and response triggered
26 cases have been reported; 20 are in isolation and receiving treatment, and six deaths have been recorded. 161 contacts of the reported cases have been identified so far and are being monitored. The Ministry of Health, Rwanda, posted a video on social media with a caption reassuring viewers that “people can continue with their daily activities” and “should not panic” as the “hotspots of the disease” have been identified.
WHO is “mobilising” expertise and outbreak response tools to “reinforce the control measures” that are being rolled out. A consignment of clinical care and infection prevention and control supplies will be delivered from the WHO Emergency Response Hub in Nairobi, Kenya, to Kigali in the next few days. Efforts are also underway to “reinforce collaborative cross-border measures for readiness and response” in countries that neighbour Rwanda.
WHO Regional Director for Africa, Dr Matshidiso Moeti, explained that the critical outbreak response aspects are being put in motion “rapidly” to “halt the spread of this virus swiftly and effectively”.
“With the country’s already robust public health emergency response system, WHO is collaborating closely with the national authorities to provide the needed support to further enhance the ongoing efforts.”
Marburg
Marburg virus disease is a “highly virulent” member of the filoviridae family and causes haemorrhagic fever. It has a fatality ratio of up to 88%, with symptoms progressing rapidly after infection. The virus is transmitted to humans from fruit bats and spread among people through direct contact with the bodily fluids of infected people, surfaces, and materials.
The disease was first recognised after large outbreaks in Germany and Serbia in 1967, associated with laboratory research involving African green monkeys from Uganda. Outbreaks and cases have been reported sporadically since then, and efforts have been made to develop effective medical countermeasures. However, there is no licensed vaccine against Marburg virus disease.
A confluence of infectious disease
The Marburg outbreak will increase pressure on the Rwandan health system, which is already fighting its mpox outbreak, declared on 27th July 2024. In September 2024, Dr Jean Kaseya, Director-General of Africa CDC, reported that Rwanda had begun an mpox vaccine campaign after receipt of 1,000 doses. The campaign targeted districts bordering the Democratic Republic of the Congo, the epicentre of the current PHEIC. How will the health services respond effectively to both infectious disease threats, and will there be similarities or ‘doubling up’ in their strategies?
For the latest infectious disease and vaccine news, don’t forget to subscribe to our weekly newsletters here. To participate in important discussions about vaccine development in outbreak situations, join your colleagues at the Congress in Barcelona next month.

by Charlotte Kilpatrick | Sep 20, 2024 | Global Health |
In September 2024, the European Medicines Agency (EMA) announced its recommendation to extend the indication of the smallpox and mpox vaccine Imvanex to adolescents from 12 to 17 years of age. Imvanex, Bavarian Nordic’s MVA-BN, is authorised in the EU for the protection of adults against smallpox, mpox, and disease caused by the vaccinia virus. The Committee for Medicinal Product for Human Use (CHMP) based this recommendation on interim results from a study comparing the effects of the vaccine in adolescents and adults.
Data-led expansion
Interim results indicate that the immune response in adolescents was comparable to adults, from which the authorities have inferred that the vaccine will “provide similar protection in adolescents to that expected in adults”. The safety profile was also comparable with no additional risk identified. EMA has requested the marketing authorisation holder to submit the results of this study by 30th May 2025 to further characterise the safety information for adolescents.
Although this is the first approval of MVA-BN as a smallpox,mpox vaccine for adolescents, Bavarian Nordic notes that a recombinant version of MVA-BN (Mvabea) received EMA approval in 2020 as part of a prime-boost regimen for the prevention of disease caused by Ebola virus in individuals aged 1 and older. This approval was based on studies involving more than 3,300 individuals, including over 800 children and adolescents aged 1-17 in Africa.
Implications for the response
EMA states that this assessment has “important implications” for the global mpox response. As EMA is the regulatory agency of record for the WHO prequalification of the vaccine earlier this month, the CHMP assessment constitutes the basis for WHO prequalification approval to “facilitate timely and increased access” in communities that need it most. The EMA’s assessment has also previously been considered by the DRC’s national regulatory authority for fast-track approval.
President and CEO of Bavarian Nordic, Paul Chaplin, applauded EMA for the “expedited” review and decision.
“This represents an important milestone in our efforts to make our vaccine available for all populations and will help improve access for some of the most vulnerable individuals mostly impacted by the ongoing mpox outbreak in Africa.”
For insights into the regulatory processes behind access to vaccines, including from senior representatives of EMA, join us at the Congress in Barcelona next month. Don’t forget to subscribe to our weekly newsletters for the latest vaccines news.

by Charlotte Kilpatrick | Sep 20, 2024 | Global Health |
In September 2024, Africa CDC offered congratulations to the Governments of Japan and the Democratic Republic of the Congo (DRC) for their agreement on the donation of Lc16 mpox vaccines and specialised inoculation needles. At a signing ceremony in Kinshasa, the two governments marked a “significant milestone” in their cooperation. These vaccines are a “critical boost” to DRC’s mpox response; Lc16 is the only mpox vaccine currently approved for children.
Lc16
The vaccine is derived from the Lister strain of vaccinia and contrasts to the replication-deficient vaccines like Modified Vaccine Ankara (MVA) by retaining most of vaccinia genome and being able to replicate at the site of inoculation. It is administered as a single dose through the scarification method, which involves scratching the skin before applying the vaccine solution. Studies in children suggested that the vaccine was safe and well tolerated, leading to licensure. However, the duration of immunity remains unclear.
Another tool for the fight
Africa CDC states that this donation comes “at a pivotal time” in DRC’s fight against the regional mpox outbreak. Since the declaration of a PHECS in August 2024, a “more coordinated international response” has evolved. Central to this response is vaccination, which is complicated by Africa’s “limited access to critical countermeasures”.
Director General of Africa CDC, H.E. Dr Jean Kaseya, is “deeply appreciative” of the ‘” of the “generous donation” of mpox vaccines and specialised vaccine needles to the DRC.
“This timely assistance will significantly bolster our ongoing efforts to contain the outbreak, and I am confident that this partnership will help mitigate the public health threat posed by mpox, not only in the DRC but across the continent.”
Japan’s Senior Deputy Minister for Foreign Affairs, Takeshi Akahori notes the increasing number of cases in the DRC and other countries, commenting that Japan is “monitoring the situation closely” with WHO.
“I hope that these vaccines and needles will contribute meaningfully to the fight against mpox.”
Join us at the Congress in Barcelona next month to engage with public health experts on current health threats and how vaccines can contribute to our response, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 18, 2024 | Global Health |
The Global Fund to Fight AIDS, Tuberculosis, and Malaria (the Global Fund) announced in September 2024 that it is responding to a request from the government of the Democratic Republic of the Congo (DRC) for support in its mpox response. The Global Fund will provide US$9.5 million towards the emergency response in six of the highest transmission provinces (Equateur, Sud-Ubangui, Sankuru, Tshopo, Sud-Kivu, and Nord-Kivu) as well as Kinshasa, which is home to 17 million people.
DRC is fighting the world’s largest mpox epidemic, reporting 5,160 confirmed cases and 25 deaths since the beginning of the year. However, as testing capacity and availability are limited, the number of suspected cases could be up to five times the number of laboratory-confirmed cases. As the epidemiology of mpox evolves in “complex ways”, implications for prevention, preparedness, and response efforts also evolve. Furthermore, mpox is “increasingly being associated with HIV”, which heightens the risk of transmission, illness, and death. Thus, investment in mpox efforts helps to strengthen work on HIV, and vice versa.
Global Fund support
The support offered by the Global Fund contributes to specific priority areas on the government’s National Preparedness and Response plan:
- Enhancing disease surveillance systems with an emphasis on strengthening early warning capabilities and alert and response systems to detect, monitor, and respond to mpox and other disease outbreaks.
- Strengthening laboratory systems and diagnostics to increase case detection and stop the spread.
- Conducting risk communication and community mobilisation and engagement through the network of community health workers and community actors who are deployed for HIV, TB, and malaria prevention and awareness.
- Implementing infection prevention and control measures to protect health workers, including at the community level.
- Reinforcing country-level coordination, planning, and support for emergency response and operations.
- Strengthening the capacity of health facilities to provide primary care services and support future emergencies.
The support also complements the current collaboration between DRC’s ministry of health, Africa CDC, WHO, humanitarian organisations, and other key partners on efforts to “address the severe challenges to the public health system” in the east of the country, where the epidemic is “converging with risks of other infectious diseases”. The Global Fund has already contributed support in moving available stocks of personal protective equipment to the most affected provinces.
Today’s fight for tomorrow’s resilience
Dr Roger Kamba, Minister of Health and Social Welfare for the DRC recalled the “proven track record” of infectious disease control in the partnerships with the Global Fund and other health partners.
“Over the past two decades, the number of AIDS-related deaths and new HIV infections in DRC have reduced by more than 60%, through coordination and collaboration across all out partners.”
Dr Kamba is “determined to continue to work in the same manner for a strong response to mpox”.
“The fight against the current mpox epidemic is a top priority for our ministry, especially through the reinforcement of the community response. It is essential to recognise that by acting now, we are not only fighting mpox but also investing in the resilience and health security of tomorrow.”
Executive Director of the Global Fund, Peter Sands, reflected that people who live in “areas of conflict and crises” often encounter “significant barriers to accessing health services”.
“When a disease outbreak occurs in these places, the challenges are compounded. Strong systems of trusted community health workers, health educators, and other local responders are essential for stopping disease spread.”
Mark Edington, Head of Grant Management at the Global Fund, emphasised the importance of swift action in disease outbreak situations.
“Immediate intervention is crucial to strengthen systems for health and improve disease detection, surveillance, and response mechanisms, aiming to prevent further deterioration in health outcomes, particularly for women, children, and internally displaced persons.”
The Global Fund encourages other affected countries to assess their mpox needs and the consider the possibility of repurposing existing Global Fund investments.
To contribute to important discussions about effective allocation of funds for emergency responses and ensuring more resilient health systems, join us at the Congress in Barcelona next month. Don’t forget to subscribe to our newsletters here for the latest vaccine and global health news.

by Charlotte Kilpatrick | Sep 18, 2024 | Global Health |
In September 2024, Gavi and Bavarian Nordic announced an advance purchase agreement (APA) to secure 500,000 doses of MVA-BN mpox vaccine to be supplied to countries in Africa that are affected by the mpox outbreak. The doses are funded by Gavi’s First Response Fund and are for delivery in 2024. Bavarian Nordic will be ready to supply the vaccine doses after a supply agreement has been signed with UNICEF, which will deliver the doses.
The First Response Fund
Gavi’s First Response Fund was established in June 2024 to “make cash rapidly available” for the purchase of vaccines in health emergencies. It is available to Gavi-eligible countries in the early days of a pandemic or major health emergency. The Fund pre-positions up to US$500 million of surge financing for vaccine procurement ‘at risk’, which means funds are used to secure doses and “Gavi’s place in the queue” while manufacturers complete the final steps of regulatory approval and manufacturing scale-up.
Dr Sania Nishtar, Gavi’s CEO, commented that the First Response Fund was designed in collaboration with donors and partners “specifically to provide rapid early funding for emergencies such as mpox”.
“Using it today to fund the first direct transaction for vaccines in support of equitable access and the global response, just over a month since mpox was declared a public health emergency, takes us a long way towards our goal of protecting those most at risk.”
Dr Nishtar thanked donors for enabling Gavi to “act rapidly” by committing funds before an emergency occurred.
“We are committed to working with affected governments and our partners to turn these vaccines into vaccinations as quickly and effectively as possible and, over time, to build a global vaccine stockpile if sufficient funding is secured for Gavi’s work through 2030.”
Paul Chaplin, President and CEO of Bavarian Nordic, is pleased to sign the agreement and “strengthen our commitment to support Gavi and other global health partners” who demonstrate “strong leadership”.
“The doses secured through this agreement will significantly increase the availability of mpox vaccines for African countries, and we are pleased that Gavi has selected our MVA-BN vaccine, which has proven highly effective during the global mpox outbreak in 2022.”
For the latest on vaccine collaborations to ensure equitable access, get your tickets to join us at the Congress in Barcelona next month, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 16, 2024 | Global Health |
In September 2024 WHO announced the establishment of an access and allocation mechanism for mpox medical countermeasures, including vaccines, treatments, and diagnostic tests. The Access and Allocation Mechanism (AAM) is intended to increase access to these essential tools for people at highest risk, ensuring that limited supplies are used “effectively and equitably”. This announcement comes after WHO declared the mpox outbreak a PHEIC in August 2024 and addresses one of the key International Health Regulations Emergency Committee’s recommendations: “equitable access to safe, effective, and quality-assured countermeasures”.
AAM
The AAM is part of the interim Medical Countermeasures Network (i-MCM-Net). Developed in response to “global vulnerabilities” exposed by the COVID-19 pandemic, i-MCM-Net enhances collaboration through a “Network of Networks” approach. It seeks to provide timely and equitable access to quality, safe, effective, and affordable medical countermeasures in response to public health emergencies through existing networks and global collaboration. The network was endorsed by WHO Member States as an interim mechanism while negotiations on a pandemic agreement continue.
The mpox AAM includes members of the i-MCM-Net as well as WHO: Africa CDC, CEPI, the EU Health Emergency Preparedness and Response Authority (HERA), FIND, Gavi, the PAHO Revolving Fund, UNICEF, Unitaid, and others. It will work to allocate the “currently scarce supplies” to those at highest risk of infection.
It will operate according to three guiding principles:
- Preventing illness and death – prioritise vaccination and other tools to interrupt transmission for those at greatest risk to prevent illness and death.
- Mitigating inequity – ensure equitable access to medical countermeasures for all people at risk, irrespective of socio-economic or demographic background.
- Ensuring transparency and flexibility – establish and maintain clear and open communication about allocation decisions and be ready to adapt strategies as new data emerge or situations change.
More than 3.6 million vaccine doses have been pledged for the mpox response, including 620,000 doses of MVA-BN pledged to affected countries by the European Commission, Austria, Belgium, Croatia, Cyprus, France, Germany, Luxembourg, Malta, Poland, Spain, and the United States of America, as well as Bavarian Nordic. Japan has pledged 3 million doses of the LC16 vaccine. This is the largest pledge so far.
International coordination
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, recognised the need for “powerful tools” like vaccines, therapeutics, and diagnostics, to bring the mpox outbreak “under control”.
“The COVID-19 pandemic illustrated the need for international coordination to promote equitable access to these tools so they can be used most effectively where they are most needed. We urge countries with supplies of vaccines and other products to come forward with donations, to prevent infections, stop transmission, and save lives.”
Dr Mike Ryan, Executive Director of WHO’s Health Emergencies Programme, emphasised that WHO and its partners are working with the government of the Democratic Republic of the Congo and other affected countries to “implement an integrated approach to case detection, contact tracing, targeted vaccination, clinical and home care, infection prevention and control, community engagement and mobilisation, and specialised logistical support”.
“The AAM will provide a reliable pipeline of vaccines and other tools in order to ensure the success on the ground in interrupting transmission and reducing suffering.”
Join us at the Congress in Barcelona next month to share your insights on the best ways to ensure equitable access to essential medical countermeasures, and don’t forget to subscribe to our weekly newsletters for the latest vaccine news.

by Charlotte Kilpatrick | Sep 13, 2024 | Technology |
In September 2024, WHO announced that the MVA-BN vaccine, manufactured by Bavarian Nordic, is the first vaccine against mpox to be put on the prequalification list. This approval is “expected to facilitate timely and increased access” to the vaccine in “communities with urgent need” amid the outbreak of mpox. The prequalification is based on information submitted by Bavarian Nordic and reviewed by the European Medicines Agency.
Recommended for use
WHO’s Strategic Advisory Group of Experts (SAGE) on Immunisation recommended the use of MVA-BN in an mpox outbreak context for persons at high risk of exposure. It can be administered in over-18s in two doses 4 weeks apart. After initial cold storage it can be kept between 2°C and 8°C for up to 8 weeks.
Although it is not currently licensed for persons under 18 years of age, the vaccine can be used “off-label” in infants, children, and adolescents, and in pregnant and immunocompromised people. This allows use in an outbreak after consideration of the potential risks in relation to the benefits of vaccination.
Data available to WHO reveal that a single-dose MVA-BN vaccine, administered before exposure, has an estimated 76% effectiveness in protecting against mpox; the two-dose schedule achieves an estimated 82% effectiveness. Post-exposure vaccination is less effective. Clinical studies have demonstrated a good safety profile and vaccine performance, which have been confirmed in real-world use during the global outbreak since 2022. However, WHO emphasises the need to collect “as much data as possible on vaccine safety and effectiveness in different contexts”.
An important step
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, described this first prequalification as an “important step in our fight against the disease” with implications for current outbreaks and the future.
“We now need urgent scale up in procurement, donations, and rollout to ensure equitable access to vaccines where they are needed most, alongside other public health tools, to prevent infections, stop transmission, and save lives.”
WHO Assistant Director-General for Access to Medicines and Health Products, Dr Yukiko Nakatani, suggested that the prequalification will “help accelerate ongoing procurement” to help communities “on the frontlines of the ongoing emergency in Africa and beyond”.
“The decision can also help national regulatory authorities to fast-track approvals, ultimately increasing access to quality-assured mpox vaccine products.”
Dr Rogerio Gaspar, WHO Director for Regulation and Prequalification, commented that the findings of the product and programmatic suitability assessments are “particularly relevant in the context of the declaration of a public health emergency of international concern (PHEIC)”.
“We are progressing with prequalification and emergency use listing procedures with manufacturers of two other mpox vaccines: LC-16 and ACAM2000. We have also received 6 expressions of interest for mpox diagnostic products for emergency use listing so far.”
To explore the steps before, during, and after these approval processes in emergency contexts, join us at the Congress in Barcelona this October. Don’t forget to subscribe to our weekly newsletters here for regular updates.

by Charlotte Kilpatrick | Sep 12, 2024 | Global Health |
Bavarian Nordic issued an update on its supply and manufacturing activities in support of mpox response efforts in September 2024. The company manufacturers the approved non-replicating smallpox and mpox vaccine MVA-BN (marketed as JYNNEOS, IMVAMUNE, and IMVANEX). The company statement describes “intensified” collaboration with global stakeholders in response to the declaration of mpox as a PHECS by Africa CDC and a PHEIC by WHO.
Progress so far
Through donations from the European Commission, the United States government, and Bavarian Nordic, initial doses of MVA-BN have been delivered to the Democratic Republic of the Congo (DRC), the epicentre of the mpox outbreak. Further donations have been pledged by other countries. However, UNICEF and Africa CDC have suggested that vaccines will be needed to protect up to 1 million people in high-risk areas of DRC, with up to 10-12 million doses required through 2025.
Bavarian Nordic “is committed to ensuring equitable access to its mpox vaccine”. It has therefore prioritised the production of MVA-BN for the rest of the year to ensure up to 2 million doses by year-end. This means that some existing orders for 2024 will be delayed to 2025 to provide “greater flexibility” to meet “additional urgent and imminent needs”. The new supply contracts include multi-year agreements with countries, including those who have pledged vaccines for Africa. Additionally, Bavarian Nordic has responded to UNICEF’s emergency tender and is participating in discussions with other organisations and individual governments around the globe.
Up to 13 million doses
By focussing “full capacity to address the current public health emergency”, Bavarian Nordic hopes to supply up to 13 million MVA-BN doses by the end of 2025. This includes 2 million in 2024. Although this may be sufficient to meet the current and near future demand, Bavarian Nordic is exploring “additional levers” to expand capacity. This includes possibly transferring manufacturing to other companies.
“Based on these early, but highly constructive discussions, together with further planned improvements in the manufacturing process, Bavarian Nordic has identified another 50 million doses that, pending regulatory approvals and demand, could be supplied during the next 12-18 months.”
President and CEO of Bavarian Nordic, Paul Chaplin, stated that the company is “working closely with all governments and organisations to support the international efforts to combat the latest public health emergency”. Mr Chaplin highlighted that Bavarian Nordic will “support all requests for vaccine and have already secured agreements to the UNICEF tender that will hopefully secure more access to MVA-BN globally”.
“We remain committed to the equitable access either through prioritising our own capacity, accelerating planned improvements in the manufacturing process and by exploring ways to further expand capacity through partnerships around the globe. Once again Bavarian Nordic, through innovation and our commitment to improving and saving lives has stepped forward as an important part of the international community’s response to the current public health emergency.”
We will hear more from Bavarian Nordic on their contributions to the mpox response at the Congress in Washington next April; get your tickets to join us there and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 11, 2024 | Global Health |
Gavi announced in September 2024 that a shipment of 15,460 doses of mpox vaccine has arrived in the Democratic Republic of the Congo (DRC) to “support the global and regional effort to contain the outbreak”. These doses were donated by manufacturer Bavarian Nordic to Gavi-eligible countries. Arriving in Kinshasa, the shipment adds to the 215,000 vaccine doses already donated by the European Union.
Global response
The DRC is the epicentre of the mpox outbreak; it has reported around 94% of all cases and 99% of related deaths. It has secured national regulatory approval of Bavarian Nordic’s JYNNEOS mpox vaccine and rollout is expected to begin shortly. Gavi has made US$2.9 million under the Fragility, Emergencies, and Displaced Populations (FED) Policy to support DRC’s vaccination efforts.
In August 2024, Gavi announced that the Board has approved the final terms of the First Response Fund. This is the “fastest tool in a suite of instruments” known as the Day Zero Financing Facility; this is intended to make resources “immediately” available for a vaccine response to an urgent public health emergency. Gavi is also working with countries and partners and sharing “legal and process knowledge” and operational support to Africa CDC and partners.
“Based on our experience working with donors, countries, and partners to coordinate the global COVAX dose donation mechanism, this includes providing information on the complex technical, legal, regulatory, and logistical considerations involved in mounting rapid vaccination campaigns with donated doses.”
CEO of Gavi, Dr Sania Nishtar, described the latest shipment as a “timely addition” to the global response.
“Now that they have arrived, our first priority is to work with our partners to ensure these and other vaccines reach those who need them, as quickly and efficiently as possible. Gavi has already unlocked funds to support this delivery.”
For regular updates on the global effort to stop the spread of mpox with vaccines, don’t forget to subscribe to our weekly newsletters here. To engage with experts working in this area, get your tickets to the Congress in Barcelona this October.

by Charlotte Kilpatrick | Sep 6, 2024 | Technology |
A paper in Cell in September 2024 presents a promising mRNA vaccine candidate against mpox disease. The mRNA-lipid nanoparticle (LNP) vaccine expresses MPXV surface proteins and was compared with modified vaccinia Ankara (MVA) vaccine, proving to confer protection against challenge and mitigate symptoms and disease duration. Furthermore, it provided “enhanced viral control and disease attenuation” compared to MVA, which highlights the potential of mRNA vaccines against future pandemic threats.
A new modality
Despite the availability of an effective vaccine against mpox, the authors note “issues in supply, unfavourable reactogenicity, incomplete immunity, and uncertainty of cross-protection”. These factors provide “critical motivation” for the pursuit of a new vaccine modality for “improved vaccines to cover these gaps”. mRNA vaccines offer “unprecedented flexibility, speed, and immunogenicity”. However, it was unclear whether an mRNA vaccine could provide “comparable immune protection” to a whole attenuated poxviral vaccine vector.
The study
The authors used a stringent clade I MPXV Zaire 1979 (Z79) MPXV nonhuman primate (NHP) model to assess the protective efficacy of mRNA-1769, an mRNA-lipid nanoparticle (LNP) vaccine. It expresses optimised versions of four antigens of interest (A29, A35, B6, and M1). This vaccine was compared with mRNA; both vaccines were administered in “clinically relevant doses”.
Both vaccines conferred “complete protection” after lethal MPXV challenge. However, mRNA-immunised animals experienced 10-fold fewer lesions, reduced disease duration, and “substantial” mitigation of circulating and mucosal viraemia. Furthermore, deep immunological profiling of the humoral response revealed more robust MPXV neutralising responses, broadly reactive heterologous neutralising titres, and greater functional humoral immune responses against the four antigens in the mRNA-immunised animals.
Immune correlates analyses highlighted the “critical coordination” between neutralising and Fc-effector functions against both EV and MV targets, EV-Fc target-specific functions and neutralisation as key correlates of antiviral control, and EV target-antigen-specific opsonophagocytic activity and neutrophil/natural killer cell-targeted functions to the MV as “key determinants” of lesional control. These results suggest that the mRNA-LNP vaccine induced a robust functional humoral response that provided protection against a lethal MPXV challenge. This is like MVA immunisation but with the benefit of “superior protection against disease”.
“These data provide critical insights into mRNA-vaccine-induced correlates of immunity against MPXV, which can support licensure, provide mechanistic insights on vaccine performance, support optimised vaccine usage in vulnerable populations, and inspire redesign should novel Orthopoxviral threats emerge requiring antigen addition or alteration.”
Rapid responses
The authors reflect on the “lack of vaccine deployment and access to medical countermeasures”, which has “fuelled the spread” of mpox from “traditionally endemic rural areas” to larger metropolitan centres. They suggest that lack of routine immunisation with contemporary VACV-based vaccines has provided “fertile ground” for low-level spread of the virus and opportunities for mutation.
“Thus, additional safe and highly efficacious vaccine platforms that are rapidly adaptable upon viral mutation are urgently needed.”
Nucleic-acid vaccines, like mRNA-LNP vaccine technologies, facilitate rapid responses to emerging viral threats. Sequences against key genes can be synthesised and converted into a potential vaccine quickly, and production can take place at regions of interest through worldwide manufacturing centres.
Join us at the Congress in Barcelona to participate in discussions about the potential of mRNA vaccines against various diseases and don’t forget to subscribe to our weekly newsletters for regular vaccine updates.

by Charlotte Kilpatrick | Sep 5, 2024 | Global Health |
Bavarian Nordic announced in September 2024 that the first doses of its mpox vaccine, MVA-BN (JYNNEOS), have arrived in the Democratic Republic of Congo (DRC) to support response efforts. Further shipments are planned within the next few days, contributing to a total of more than 250,000 doses donated by the European Commission’s Health Emergency Preparedness and Response Authority (HERA), the United States government, and Bavarian Nordic.
First doses arrive
This shipment brings the first mpox vaccines to the DRC, which Bavarian Nordic describes as a “turning point” in the joint efforts by Africa CDC and the international community. The DRC authorities have issued national emergency use authorisation for MVA-BN to allow immediate deployment. Broader deployment on the continent is “pending an emergency use listing” from WHO.
WHO Director-General Dr Tedros Adhanom Ghebreyesus shared his reaction to the delivery on social media, emphasising that WHO and partners are supporting DRC in the delivery of a “comprehensive health emergency response based on case finding, contact tracing, targeted vaccination, case management, and community engagement and mobilisation”.
“Once again, we are grateful to the European Union for its solidarity and for sharing vaccines.”
Responding to the call
DRC is the “epicentre” of the African mpox outbreak, reporting more than 94% of reported cases and nearly 99% of the reported deaths year-to-date on the continent. Although surveillance and control are “continuously improving” the true burden of disease remains unclear as case numbers are “underreported”. Bavarian Nordic’s President and Chief Executive Officer, Paul Chaplin commented on the “alarming rate” of increased cases in the DRC, where “aid is desperately needed”.
“Africa CDC has called for the international community to step up and mobilise the resources needed to combat the outbreak, and we are proudly responding to this call together with our partners in the European Commission’s Health Emergency Preparedness and Response Authority and the U.S. government, who have demonstrated strong leadership and determination during this serious health crisis.”
Mr Chaplin stated that “time is of the essence” but is pleased that “the way has now been paved for our mpox vaccines to reach the people in the DRC who are most in need”.
“We will continue our collaborative efforts with both international partners and local authorities to further broaden access to our vaccine in the region.”
While this news is welcomed by public health experts, will the doses be enough to control the outbreak, and is there a risk of perpetuating dependence on donations? For more on preparedness and response efforts at the Congress in Barcelona this October, get your tickets now. Don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 5, 2024 | Global Health |
In response to the mpox outbreak, declared a PHEIC by WHO and a PHECS by Africa CDC, the two organisations announced that they are co-leading a “coordinated, continent-wide response”. The Mpox Continental Preparedness and Response Plan for Africa describes “essential priorities” to control the current outbreak, focusing on ten pillars. The plan categorises Member States into four risk-based groups to ensure efforts and resource allocation are targeted. The estimated budget for September 2024 to February 2025, excluding the cost of vaccines, is US$599,153,498
Collective commitment
In the foreword by Africa CDC Director General Dr Jean Kaseya and WHO Africa Regional Director Dr Matshidiso Moeti, the declaration of mpox as a PHECS is described as a “bold move”. This was followed by WHO’s declaration, reflecting “alignment” and “collective commitment to raising awareness, mobilising resources, and galvanising action at all levels”. Drs Kaseya and Moeti state that current “battle” against mpox has been shaped by “hard-earned lessons” from the COVID-19 pandemic.
“The experience of COVID-19 exposed vulnerabilities in our health systems, showed Africa’s inequity and unfair treatment in terms of access to medical countermeasures, highlighted the urgent need for enhanced preparedness, and underscored the importance of swift, coordinated action in the face of emerging health threats.”
The “foundation” of the mpox response is built on lessons of “solidarity, resilience, and collaboration”.
4-ONE
A new approach is outlined: a “4-ONE APPROACH”:
- ONE coordination mechanism
- ONE continental response plan
- ONE budget
- ONE monitoring and evaluation mechanism
Africa CDC and WHO will lead efforts to implement the “unified approach” with global and continental stakeholders. The plan is a “roadmap” to facilitate a “coordinated, comprehensive, and evidence-based response” that puts the principles of “equity, inclusivity, and accountability” at the centre.
“As we move forward, we are guided by our strong commitment to protecting the health of all Africans, enhancing our collective resilience, and securing a healthier future for our continent. Together, we will overcome this challenge and build a stronger and resilient Africa.”
Mpox: then and now
Mpox was first described in the Democratic Republic of Congo (DRC) in 1970. It is a viral zoonotic illness that has caused “numerous outbreaks” since its identification. Although early outbreaks tended to be associated with zoonotic transmission from wildlife to humans, recent cases in urban settings have suggested changes in transmission dynamics.
“The emergence of zoonotic diseases is driven by complex ecological, climatic, political, economic, security, and social factors, some of which are becoming further exacerbated on the continent.”
However, the “warning signs” of local outbreaks are often “neglected” with “limited investigation, surveillance, diagnosis, and response”. Despite improvements in surveillance and reporting systems to enhance the understanding of mpox’s epidemiological patterns, “significant gaps” remain. Mpox virus has two variants: clade I and clade II. Clade I is geographically concentrated around the Central and Eastern Africa region and is considered “more virulent”; Clade II is found in Western Africa and other regions.
In the global outbreak of 2022-2023, the disease spread drew “renewed focus” on medical countermeasures. While many countries outside Africa were “quick to respond”, Africa faced “significant challenges in accessing these crucial tools”. Despite the high burden of mpox in several countries in Africa, access to vaccines and other medical countermeasures was inequitable.
“This lack of access was due to multiple factors, including limited global production capacity, unequal distribution agreements, and a lack of investment in public health infrastructure in Africa.”
Vaccines like JYNNEOS (MVA-BN) and ACAM2000 were widely authorised for emergency use but were “largely unavailable to African countries”. The authors of the plan attribute this to pre-existing contracts between manufacturers and high-income countries. Furthermore, logistical challenges exacerbated the disparity; “inadequate” cold chain storage facilities and distribution networks” created obstacles to the delivery of countermeasures.
“This inequity underscored the urgent need for Africa to develop self-reliance in manufacturing and distributing medical countermeasures to avoid similar scenarios.”
The current situation is concerning; reported cases are increasing in number across the continent. In comparison with 2022, there was a 79% increase in reported cases in 2023. By 3rd September 2024, confirmed cases have exceeded the number reported in 2023 by over 3,700. Furthermore, the recent outbreak has “dramatically” affect children under 15 years (60%). In 2024, 13 countries have reported cases, with a new subvariant of mpox clade I (clade Ib) identified since September 2023. This has been ‘widely circulating” among commercial sex workers and their sexual contacts.
While the increasing cases are worrying, the “true burden” is uncertain. Thus, the authors demand enhanced surveillance and detection. They also highlight the need for vaccination of both targeted and expanded priority population groups, particularly in the context of Africa’s “weaker surveillance systems and limited diagnostic capacity”.
“The Mpox Continental Preparedness and Response Plan for Africa (MCPRPA) seeks to build a stronger foundation for health security in Africa through a country-driven unified approach, prioritising prevention, enhancing immunity at community level, and promoting the continent’s self-reliance.”
Risk categories
The plan classifies African Union Member States according to their mpox status and risk level. The risk level is for “planning and resource optimisation”.
- Experiencing sustained human-to-human transmission: DRC, Burundi, Nigeria, South Africa, Côte d’Ivoire, Central Africa Republic
- Not already falling into category 1 but experiencing sporadic human cases since 1st January 2022 and/or countries that are assessed as having endemic zoonotic reservoirs for mpox: Rwanda, Kenya, Uganda, Sierra Leone, Libera, Ghana, Cameroon, Gabon, Republic of Congo, Morocco, Egypt, Benin, Mozambique, Sudan
- Not already falling into the first two categories that are assessed as requiring readiness including due to proximity to category 1 countries by land, air, or sea: Angola, Zambia, Eswatini, Lesotho, Ethiopia, South Sudan, Tanzania, Malawi, Republic of Guinea
- All other countries
Guiding principles
The plan relies on guiding principles from lessons learnt during the COVID-19 pandemic; the align with the 2023 Lusaka Agenda, which emphasises “strengthening joint approaches for achieving equity in health outcomes, operational coherence, and a coordinate approach to product development and research”.
- Country-driven: The plan focuses on mpox preparedness and response interventions based on priorities identified by affected countries to ensure that the response is tailored to the needs of each country.
- Science-driven: The strategic approaches and key interventions are grounded in the best available scientific evidence, ensuring that the response is effective and adaptive to the evolving understanding of the virus and its transmission.
- Equity and solidarity: Prioritisation of issues and resource allocation should be sensitive to the needs of the most affected regions/provinces, vulnerable groups, and countries most in need. This is supported by global solidarity, ensuring that medical countermeasures are made available to African Member States equitably.
- Unified: Align all partners around a single cohesive plan, ensuring that all stakeholders work toward common objectives, minimising duplication and maximising impact.
- Single collaboration mechanisms: Streamline efforts through coordinated leadership.
- Sustainability: Focus on developing sustainable, long-term solutions that can be scaled and maintained over time, ensuring that countries are better prepared for future outbreaks and that response efforts have a lasting impact.
10 pillars
The plan has 10 pillars, each with a strategic objective and actions.
- Coordination and leadership
- Strategic objective – establish one functional coordination mechanism with one team, one plan, one budget, and one monitoring and evaluation (M&E) framework at continental, national, and subnational levels.
- Actions – enhance harmonised coordination and collaboration between relevant stakeholders including resource mobilisation.
- Risk communication and community engagement (RCCE)
- Strategic objective – support and engage communities, particularly the most vulnerable members, so that they practice key public health recommendations and access the needed services to reduce transmission, morbidity, mortality, and secondary impacts.
- Actions – engage communities in public health response and ensure their perspective and realities drive the mpox response interventions.
- Surveillance
- Strategic objective – establish/enhance functional event-, community-based-, and cross-border mpox surveillance systems at continental, national, subnational levels.
- Actions – strengthen mpox surveillance through event/community-based surveillance, contact tracing, point of entry, and cross-border information sharing.
- Laboratory capacity
- Strategic objective – strengthen mpox laboratory testing and sequencing capacity to confirm at least 80% of suspected mpox cases and sequence at least 5% of epidemiologic and geographic representative confirmed mpox cases.
- Actions – strengthen laboratory testing for diagnostic and sequencing through training and provision of equipment and reagents.
- Case management
- Strategic objective – support comprehensive case management for mpox, including medical, nutritional, and psychosocial care, to reduce the case fatality rate to below 1% (0.5%).
- Actions – strengthen case management for mpox.
- Infection prevention and control
- Strategic objective – strengthen infection prevention and control measures at 80% of health facilities and schools in hotspots of mpox-affected and at-risk Member States to minimise the risk of mpox transmission.
- Actions – strengthen infection and prevention control measures at households, schools, health facilities, and communities.
- Vaccination
- Strategic objective – support the administration of mpox vaccination to 80% of the targeted population.
- Actions – vaccination of targeted and expanded high-risk population groups is a proactive measure to address the delayed responses that can occur due to weaker health systems, weaker surveillance systems, and limited diagnostic capacity. This would build population resilience, reduce the public health impact of mpox, and prevent healthcare systems from becoming overwhelmed. Mpox vaccination will be implemented in two phases. In the first phase, vaccines will be administered to the exposed group of contacts and the contacts of contacts and the expanded group of those at risk. In the second phase, consideration could be given for affected communities, depending on progress in epidemiology and vaccine availability.
- Research and innovation
- Strategic objective – coordinate and conduct mpox operational and clinical research across the continent to address critical knowledge gaps and support response efforts, and coordinate and enhance research and development (R&D) for the manufacturing of countermeasures to ensure rapid deployment during outbreaks.
- Operations support and logistics
- Strategic objective – provide robust operational support, ensuring the safety and security of response staff, maintaining key infrastructure and ensuring the efficient procurement and distribution of essential supplies.
- Actions – ensure robust support by developing standards for mpox supplies, coordinating demand forecasts, enhancing supply transparency and implementing fair allocation, strengthening logistics, and maintaining supply chain integrity for equitable distribution.
- Continuity of essential services
- Strategic objective – advocate for and support Member States to monitor the implementation of basic services ensuring continuity to avert loss of gains.
Budget
The plan also details “key resource requirements” for the first six months of operations. The estimates assume an initial case load of 2,000 cases per week, which increases to 4,000 cases per week in the first two months of operations. This is expected to continue through the fourth month, after which cases might decrease. The total estimated number of suspected cases is 92,000 over the first six months. Vaccine procurement costs are excluded from budget estimates as these depend on the outcome of “ongoing negotiations” with manufacturers.
The overall estimated budget for the six-month plan is US$599,153,498. Of this, 53% (US$315,311,463) are assigned to mpox outbreak response effort in the 13 affected Member States. 2% (US$14,000,000) will support the 15 high risk, non-affect Member States with emergency preparedness and 45% (US$269,842,035) will go toward partners’ operational and technical support.
Monitoring and evaluation
The monitoring and evaluation of the plan are centred on a results-based management approach, ensuring capture and analysis of key performance results information and dissemination for management decision-making, reporting, and stakeholder use.
- Input and output monitoring will be ensured through reporting tools developed by the incident management system (IMS). Periodic and ad-hoc joint support supervision visits will take place and internal review mechanisms will be used to ensure the correctness, completeness, and timeliness of monitoring data.
- The Continental incident management team (IMT) will conduct periodic evaluations of the plan.
- Data collection will be shared with the Continental IMT, which has the primary mandate for its monitoring.
Will this approach be sufficient to control the outbreak and establish mechanisms for future health threats on the continent? For expert insights into equitable vaccine development and deployment, get your tickets to join us at the Congress in Barcelona this October, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 3, 2024 | Global Health |
A study in Vaccine X presents a “comprehensive view of people’s opinions, fears, and behaviours” about mpox. The authors searched various sites for descriptive cross-sectional study designs from 2022 and 2023 addressing “knowledge, attitude, perception, preparedness, willingness to get vaccinated, and practices” against mpox infection. They conclude that there is a need to increase knowledge about mpox and spread awareness on the importance of preventive measures like vaccination.
Awareness, attitudes, and actions
“The public’s response to an epidemic is influenced by each person’s perceptions of the illness and their ability to change their behaviour as conditions change.”
The paper describes prevention and treatment of mpox as “challenging” in areas where it is endemic. Prevention measures include isolation and immunisation, with WHO recommending the use of MVA-BN or LC16 vaccines, or the ACAM2000 vaccine “when the others are not available”. Further preventive actions require “good understanding of the nature of the virus”. For example, environmental surveillance can identify the spread of pathogens within societies, shedding light on possible pathways of transmission.
A key challenge in controlling the spread of Mpox is a lack of knowledge in healthcare workers (HCWs).
“Therefore, there must be good awareness and appropriate attitudes and actions toward mpox among the HCWs and the general population.”
The study
The study was intended to provide an overview of “knowledge, attitudes, willingness to get vaccinated, level of awareness, worry, and perception of risk” among different populations. The researchers searched several databases in line with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards. Eligibility criteria included studies in any language, descriptive cross-sectional study designs conducted in 2022 and 2023, and studies addressing knowledge, attitude, perception, preparedness, willingness to get vaccinated, and practices against mpox infection. Studies measuring these outcomes were included through validated questionnaires. An overall score was calculated to determine the knowledge, attitude, perception, preparedness, worries, and practices as well as willingness to get vaccinated.
The search resulted in a total of 493 articles, reduced to 289 after the exclusion of duplicates. 37 articles were included in full-text screening, resulting in 30 eligible articles for the systematic review and meta-analysis. These were cross-sectional studies featuring healthcare workers (14), general population (10), medical students (4), and university students in different specialities (2).
Findings
The authors describe knowledge of the disease, attitudes to prevention, and intentions to follow advised practices as “major determinants” of the adoption of preventive measures. For many diseases for which vaccines currently exist, higher vaccination rates are “very important” to generate higher immunity rates. To create necessary demand, the authors highlight the need to develop safe and effective vaccines but also to “ensure that the necessary logistical issues, equitable distribution, and the population acceptance are addressed”.
The results show that “less than half had good knowledge, while the majority had good attitudes toward mpox”. Most respondents were willing to take the mpox vaccine, and the majority were reported to be aware of mpox. Less than half had worries and perception of risk toward mpox, and knowledge was highest in the general population, followed by HCWs. However, HCWs were the “most willing population” to be vaccinated against mpox.
Different characteristics had influences on knowledge about mpox but not always to the same effect; for example, “gender may have an impact on knowledge levels, but the direction of that influence may be determined by other factors, such as cultural or societal standards”. Although some studies suggested that “higher education levels and certain professional backgrounds were related with more knowledge”, professional experience was not always associated with high knowledge levels.
65% of participants expressed willingness to get vaccinated, which “can’t be considered a high percentage” as herd immunity demands more than 80% of population vaccination. A study that reported only 8.8% willingness for vaccination attributed this to “inadequate levels of factual knowledge”. Factors associated with not getting vaccinated were “various” including gender, age, income level, and education level.
Less than half of the participants were reported to be worried about mpox (42.7%), but one study found 33.2% of participants were more about mpox than COVID-19. Another study reported that male HCWs were “less worried” about mpox than females, medical students were “significantly more worried” compared to other participants, and HCWs who had experienced COVID-19 infection were “significantly less worried” about the mpox outbreak.
Conclusion
The paper concludes that mpox knowledge should be increased through awareness campaigns and social media. The importance of advice to take mpox vaccines is also highlighted, with a focus on vaccine efforts in “vulnerable groups”.
“If adequate management and prevention strategies are implemented in the early steps, the virus will be controlled adequately.”
After the declaration of mpox as a PHEIC in August 2024, how might these attitudes change? Do you think the study can be used to inform public awareness campaigns and vaccination drives? For more on encouraging vaccine confidence at the Congress in Barcelona this October get your tickets here, and don’t forget to subscribe to our weekly newsletters for the latest vaccine updates.

by Charlotte Kilpatrick | Sep 2, 2024 | Global Health |
UNICEF announced in August 2024 that it has issued an emergency tender for the procurement of mpox vaccines amid the public health emergency declared by Africa CDC and WHO. UNICEF is the world’s largest single vaccine buyer, reportedly procuring “more than 2 billion” doses annually for routine child immunisation and outbreak response on behalf of almost 100 countries. The tender is to help secure mpox vaccines for the “hardest hit countries” in a collaboration with Africa CDC, Gavi, WHO, PAHO, and other partners.
“This collaboration to increase access and timely allocation also includes working to facilitate donations of vaccines from existing stockpiles in high-income countries with the aim of containing the ongoing transmission of mpox.”
Emergency tender
Through the emergency tender UNICEF will establish conditional supply agreements with vaccine manufacturers to purchase and ship vaccines “without delay” once countries and partners have secured financing, confirmed demand and readiness, and regulatory requirements are in place. WHO is reviewing the information submitted by manufacturers in response to its invitation for expressions of interest for Emergency Use Listing.
The emergency tender is intended to secure immediate vaccine access and expand production. Agreements for up to 12 million doses through 2025 could be put in place, depending on demand, production capacity, and funding.
Ensuring equitable access
Director of UNICEF Supply Division, Leila Pakkala, highlighted the “paramount importance” of addressing the mpox vaccine shortage and delivering vaccines to “communities who need them now”.
“There is also a pressing need for a universal and transparent allocation mechanism to ensure equitable access to mpox vaccines.”
Dr Jean Kaseya, Director General of Africa CDC, agreed that “timely procurement and distribution” is “crucial to protecting the most vulnerable populations”. Dr Kaseya described the emergency tender as a “critical step forward in our collective effort to control the spread of this disease”.
“Africa CDC is committed to ensuring that vaccines are allocated swiftly and equitably across the continent, in partnership with UNICEF, Gavi, WHO, and other key stakeholders. Our unified response is essential to curbing the impact of this public health emergency and safeguarding the health and well-being of our communities.”
Dr Derrick Sim, Gavi’s interim Chief Vaccine Programmes and Markets Officer, echoed the significance of the emergency tender.
“Securing access to supply and financing, delivering doses, and in parallel ensuring countries are ready to administer them, are all vital actions that need to be conducted rapidly but thoroughly, and in a coordinated manner.”
Dr Sim welcomed the tender as “another positive step” in the response. WHO’s incidence manager for the global mpox response and acting Director for Epidemic and Pandemic Preparedness and Prevention is Dr Maria Van Kerkhove, who stated that a “swift, coordinated, and equitable response is critical” in the control of this mpox emergency and future iterations.
“All of us must act decisively now or risk allowing mpox to spread further and become an even greater global threat. In an interconnected world, the fight against mpox – as with other infectious diseases and health threats – cannot be waged alone.”
Dr Van Kerkhove shared that WHO is “glad to partner” on efforts to “get life-saving tools to people in need”.
For more on effective access strategies at the Congress in Barcelona this October, get your tickets to join us here, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Aug 30, 2024 | Global Health |
The Annenberg Public Policy Centre (APPC) shared a report in August 2024, revealing that the number of Americans believing COVID-19 vaccination misinformation has risen and their “willingness” to take or recommend vaccination against COVID-19 is “lower than in the past”. The Annenberg Science and Public Health (ASAPH) Knowledge Monitor tracks national levels of health knowledge and misinformation to generate “indices of knowledge” about health topics. The latest report is based on 20 waves of a nationally representative panel survey of US adults, the most recent of which was conducted in July 2024.
Confidence levels
The survey asks respondents to report their level of confidence in people who provide public health information. Respondents had the most confidence in primary care providers regarding “matters of public health” in 2023 and 2024. However, they had less confidence in public health institutions like the FDA and CDC. Respondents expressed least confidence in Dr Fauci, who stepped down as NIAID Director at the end of 2022.
In February 2024, Americans reported trusting scientists and police officers to act in their best interests “more than other groups”, including business leaders and journalists. Medical scientists were trusted “significantly more than any other group”. Confidence in the trustworthiness of the FDA exceeded specific measures of confidence concerning the FDA’s vaccine approval process. The four items assessing the FDA protecting the vaccine process from outside influence were the most highly correlated with each other and general confidence in the FDA.
COVID-19 misinformation and vaccines
The report emphasises that vaccines are “one of the great success stories of public health”. However, recent years have seen “declines in Americans’ perceptions that a variety of vaccines are safe and effective”. Although “most respondents” report vaccines as safe (65%-81%) and effective (61%-83%), respondents showed “significant declines” in perceptions of safety for MMR and COVID-19 vaccines, and in perceptions of efficacy for MMR, seasonal flu, and pneumonia vaccines.
Respondents considered MMR and seasonal flu vaccines safer and more effective (75%-83%) than the COVID-19 (65%-66%), even though CDC evidence indicates that the COVID-19 vaccines are “actually more effective” than flu vaccines. The authors also identify an increase in perceptions that the COVID-19 vaccines are “very or somewhat unsafe” (18%-24%).
The surveys tracked the amount of endorsement of five COVID-19 vaccine misinformation beliefs for nearly three years. Although most respondents still endorse the “science-consistent response” (55%-65%), endorsing the “science-inconsistent response” has increased over time. The “misinformed belief” that COVID-19 vaccinations have been responsible for thousands of deaths in the US increased from 22% in June 2021 to 28% in July 2024. Another trend was an increase in the “false belief” that it is safer to get a COVID-19 infection than a COVID-19 vaccine.
Vaccination in pregnancy
From June 2023 to April 2024, respondents increased their understanding of the vaccinations recommended during pregnancy by the CDC. In the most recent assessment, many respondents knew that seasonal flu (50%), COVID-19 (43%), and the Tdap (35%) vaccines are recommended in pregnancy. However, the recent survey also found that “large numbers of people” are “uncertain or do not know” the benefits of COVID-19 vaccination during pregnancy. Opinions were divided on whether to recommend the RSV vaccine to a pregnant friend or family member.
Measles
Despite the availability of an MMR vaccine that provides “long-lasting protection” against measles for people who have received both recommended doses, only 93% of kindergarten students in the US in 2022-2023 had received both doses. Exemption requests in the 2022-2023 school year, while still low, increased to 3.0% from 2.6% in the previous year.
“These increases in exemptions could be attributable to actual increases in vaccine hesitancy or persistent barriers to vaccination for families whose access to routine childhood vaccination series was reduced by the COVID-19 pandemic.”
The American public “remains relatively confident” in the vaccine for measles, mumps, and rubella. In October 2023, respondents perceived the MMR vaccine as “safer and more effective than any other surveyed vaccine”; 81% reported that the MMR is either “somewhat or very safe” and 83% reported it as “somewhat or very effective”. However, these perceptions represent a “significant decline” from August 2022, when 88% of respondents reported that the MMR vaccine was “somewhat or very safe” and 87% perceived it as “somewhat or very effective”.
In April 2024, a “large proportion of the public” knew that medical professionals recommend taking the MMR vaccine. However, less than half of respondents (49%) know that it is not more harmful than helpful to give children more than a single vaccine on the same day, and many were “not sure” (23%). Indeed, combining vaccines reduces the overall number of visits to the doctor, reducing barriers to “full, on-time vaccination”. Only 63% of respondents believe that healthy children should meet school vaccination requirements for attendance in public schools.
Most respondents (56%) were unsure about the effect of measles on potential pregnancy complications. About 4 in 10 people correctly identified two complications associated with contracting measles while pregnant: delivering a low-birth-weight baby and early delivery. Some people incorrectly indicated that diabetes (7%), blurred vision (11%), and death (12%) are more likely to occur if measles is contracted during pregnancy; this is not the case. Of particular concern is that a quarter of US adults still do not know that there is “no causal evidence” linking the measles vaccine to autism.
Mpox
As the “salience” of mpox receded in the US after the 2022 global outbreak, so has the public’s knowledge concerning the issue. The public is “significantly less worried about contracting mpox”; only 5% of respondents reported being “somewhat or very worried” about contracting mpox in the next 3 months. In July 2024, only 9% were worried about personally contracting mpox or someone in their family contracting mpox. 76% of respondents reported in October 2022 that they were “very likely or somewhat likely” to receive an mpox vaccine if they were exposed.
“In the immediate aftermath of the 2022 global mpox outbreak, many in the public learned important public health knowledge to help prevent and treat the disease. With new outbreaks recently declared in Kenya and the Central Africa Republic, now is the time for public health officials to remind the public of the risks, symptoms, and means of treatment.”
STIs
Sexually transmitted infections (STIs) are “on the rise” in the US. Thus, it is “not surprising” that 47% of respondents reported either having personally been diagnosed or knowing someone who had been diagnosed with an STI. However, just over half of respondents (54%) know that a case of syphilis can be permanently cured and most either believe (mistakenly) that there is a vaccine to prevent it (16%) or are unsure (45%). The public is “not sure” whether some STIs can be permanently cured or whether a vaccine exists to prevent them.
When asked about vaccines to prevent these infections, 67% of the public are aware that these a vaccine for HPV. 44% know that there is a vaccine for mpox. For infections without a vaccine, most of the public is either unsure or incorrect about whether that is the case:
- 61% of people do not know there is no vaccine for syphilis
- 52% of people do not know there is no vaccine for HIV
- 57% of people do not know there is no vaccine for gonorrhoea
- 55% of people do not know there is no vaccine for genital herpes
- 59% of people do not know there is no vaccine for chlamydia
To read the full report click here. Get your tickets to join us at the Congress in Barcelona for discussions about vaccine confidence, public health communication, and vaccine uptake, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Aug 30, 2024 | Technology |
In August 2024, Emergent BioSolutions announced that the US FDA has approved the supplemental Biologics License Application (sBLA) for the expansion of the indication for ACAM2000 to include prevention of mpox disease in individuals determined to be at high risk for mpox infection. The approval draws on previous human safety data and animal data, where ACAM2000 was shown to be effective in protecting against mpox virus exposure. ACAM2000 is a single-dose vaccine administered percutaneously through a bifurcated needle.
ACAM2000
ACAM2000 is the primary smallpox vaccine designated for use in a bioterrorism emergency; it is indicated in the US for active immunisation for the prevention of smallpox and mpox disease in individuals determined to be at high risk for smallpox and mpox infection. It was first approved by the FDA in 2007 for active immunisation for the prevention of smallpox disease in individuals determined to be at high risk for smallpox infection. The FDA approval follows the announcement that Emergent has filed an Expression of Interest (EOI) with WHO for consideration for Emergency Use Listing.
Emergent’s president and CEO Joe Papa commented that the FDA approval “further strengthens and broadens” Emergent’s “industry-leading smallpox portfolio”.
“This expanded indication for ACAM2000 comes at a critical time as the global health community comes together to ensure an effective and cohesive response to the recent upsurge in mpox cases. We believe Emergent is poised to support the global response needed by actively engaging with world health leaders, as well as deploying product currently available in inventory based on the needs, as well as the ability to increase supply.”
Dr Amesh A. Adalja, FIDSA FACP FACEP, health security and emerging infectious diseases expert, Johns Hopkins Centre for Health Security, described the “enormous need to use all effective tools to extinguish” the “uncontrolled epidemic” of mpox.
“ACAM2000, a direct descendent of the Jenner vaccine (humanity’s first) which was used to eradicate smallpox, and now with the broadened indication, will be an invaluable tool in this endeavour.”
For more on the vaccine community’s efforts to address the growing threat of mpox, join us at the Congress in Barcelona this October. Don’t forget to subscribe to our weekly newsletters for vaccine updates here.

by Charlotte Kilpatrick | Aug 27, 2024 | Technology |
Tonix Pharmaceuticals announced in August 2024 that it is collaborating with Bilthoven Biologics (BBio) to advance TNX-801. TNX-801 is Tonix’s mpox vaccine candidate, a live replicating, attenuated virus vaccine based on horsepox. It has demonstrated better tolerability than 20th century vaccinia viruses in animal models, and preclinical studies have shown “positive” efficacy data.
TNX-801
Tonix’s TNX-801 is in preclinical development for the prevention of mpox and smallpox. In preclinical studies, Tonix found that TNX-801 vaccination protected non-human primates against lethal challenge, prevented clinical disease and lesions, and decreased shedding in the mouth and lungs. It is delivered percutaneously as a single dose, which could enable “higher rates of community protection”.
“TNX-801 has the potential to make a global impact on mpox and the risk of smallpox because of its durable T-cell immune response, the potential to manufacture at scale, and the use of a lower dose than non-replicating vaccines.”
Bolstering preparedness and response
Jurgen Kwik, Chief Executive Officer at Bilthoven Biologicals, believes that the current mpox outbreak “exemplifies precisely why we built the pandemic preparedness facility at BBio”.
“The establishment of the ‘ever-warm’ facility for pandemic preparedness underscores the critical importance of readiness in the face of global health emergencies, such as mpox. This collaboration encapsulates the essential role of the facility in bolstering pandemic preparedness and response capabilities.”
Dr Seth Lederman, Chief Executive Officer of Tonix Pharmaceuticals, looks forward to collaborating with BBio and “accelerating the development of our vaccine candidate to prevent mpox”. Dr Lederman believes that the single dose administration will “improve acceptance and eliminate partial vaccination” when compared to the current two-dose regimens.
“We believe TNX-801 can be rapidly scaled up for manufacturing and can be distributed and stored without a costly and cumbersome ultra-cold supply chain.”
Dr Lederman highlighted that the “worldwide availability of an affordable, safe, and effective single dose” vaccine against mpox is “essential given the pandemic potential of the disease”. Furthermore, successful development of TNX-801 would “establish the foundation” for potential expansion of the viral vector platform.
We look forward to learning more about this vaccine at the Congress in Barcelona this October; get your tickets to join us there and don’t forget to subscribe for more vaccine updates here.