by Charlotte Kilpatrick | Sep 18, 2024 | Technology |
MinervaX and Wacker Biotech announced a collaboration to manufacture active protein ingredients for MinervaX’s Group B Streptococcus (GBS) vaccine in September 2024. The vaccine candidate is a novel protein-only vaccine, based on fusions of “highly immunogenic and proactive protein domains” from selected GBS surface proteins. Wacker Biotech is to manufacture the active ingredients and perform technology transfer, process validation, and process characterisation activities for commercial manufacturing. The organisation will also perform key functions to ensure “stable commercial supply” at its Amsterdam site.
GBS
Group B Streptococcus (GBS) is a “leading cause” of neonatal and infant sepsis and meningitis. The highest incidence is in neonates and young infants up to 3 months old. Between 15% and 25% of the population are “spontaneously colonised” with GBS, but during pregnancy this carries the risk of transmitting the bacteria to the child in the womb, during birth, and/or during the first months of life. WHO emphasises the need for an effective maternal GBS vaccine to protect neonates and infants before they can elicit and effective immune response.
MinervaX identifies an “unmet medical need” as there is no “general implemented and fully protective preventative treatment” for GBS. Furthermore, older adults and adults with some co-morbidities are at increased risk of severe GBS infections. This population would also benefit from a prophylactic vaccine.
A protein approach
MinervaX has developed a novel protein-only vaccine based on fusions of “highly immunogenic and protective protein domains” from the Alpha-like protein family (AlpN). This approach is intended to deliver a vaccine with “broad coverage and protection”. The lead candidate, AlpN GBS, comprises two fusion proteins, each containing two AlpN-terminal domains: GBS-NN (containing RibN and AlpCn) and GBS-NN2 (containing Alp1N and Alp2/3N).
Two Phase II clinical trials of the maternal vaccine have been successfully completed, and MinervaX is preparing for Phase III trials in this indication. Data are “very positive”, with an “acceptable” safety profile in pregnant participants and their infants, and “high immunogenicity”.
Joining forces
Dr Per Fischer, CEO of MinervaX, commented that GBS “can be life-threatening for newborn babies” and is associated with “over half a million preterm births annually”. However, after €54 million financing last year, the team is advancing the development of their novel prophylactic vaccine “for the benefit of all populations at risk, worldwide”.
“Wacker Biotech is a robust manufacturing partner with a strong track record in late clinical and commercial supply and we look forward to collaborating with the team ahead of commencing Phase III studies.”
Managing Director of Wacker Biotech B.V in Amsterdam, Ronald Eulenberger, looks forward to bringing Wacker Biotech’s strengths to the collaboration.
“With our strong background in E. coli processes, process characterisation, and process validation experiences, we at Wacker Biotech are perfectly suited to support MinervaX with its ongoing programme for the prevention of invasive GBS disease.”
We look forward to hearing from MinervaX’s CSO, Dr Bengt Johansson Lindbom, on a panel exploring the role of vaccines and mAbs in neonatal sepsis prevention at the Congress in Barcelona next month. Get your tickets to join us there and don’t forget to subscribe to our weekly newsletters for the latest vaccine news.

by Charlotte Kilpatrick | Mar 13, 2024 | Global Health |
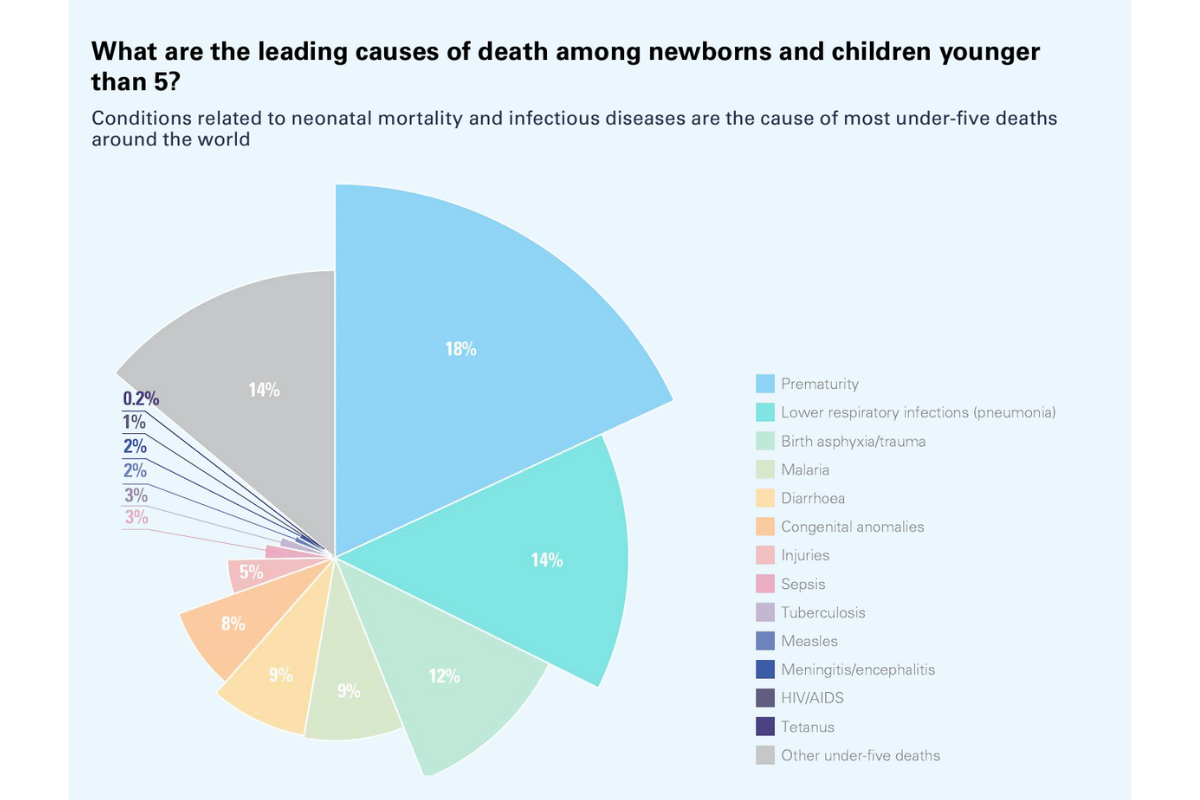
In March 2024 the United Nations Inter-agency Group for Child Mortality Estimation (UN IGME) shared a report that indicates that the number of children who died before their fifth birthday reached a “historic low” in 2022. The number dropped to 4.9 million, which has been described by health leaders as “progress”. However, there is evidently more to be done to ensure that more children survive early childhood and go on to live healthy and happy lives.
8 facts
The report begins with “8 things to know about newborn, child, adolescent, and youth mortality”.
- Good progress has led to fewer children dying before turning 5 than ever recorded.
- The annual death toll among children, adolescents, and youth remains unacceptably high.
- Children endure unequal chances of survival based on where they live, their socio-economic group, and if they live in a fragile and conflict-affected setting.
- Unless urgent action is taken to end preventable newborn and child deaths, many low- and lower-middle-income countries will not meet the Sustainable Development Goal (SDG) targets for newborn and under-five mortality and 35 million under age 5 will die before 2030.
- Progress is possible, even in low- and lower-middle-income countries.
- Investments at local, subnational, and national levels must be made to ensure proven interventions are available and accessible in every community, particularly where newborns and children are most at risk.
- Data are least available in the places where children face the highest risks.
- Global action is needed now to ensure progress – not complacency – defines the remaining years of the SDG era.
Data highlights from the report
- In 2022 neonatal deaths reached 2.3 million, which means one death every 14 seconds.
- In 2022 deaths of children under 5 reached 4.9 million, which means one death every 6 seconds.
- In 2022 adolescent deaths reached 0.9 million, which means one death every 35 seconds.
Comparing previous recorded years, the report tracks total deaths for earlier decades. From 1990-1999 there were 115 million deaths; from 2000-2009 there were 85 million deaths; from 2010-2019 there were 62 million deaths. Between 2020 and 2022 there have been 15 million deaths.
The risk of death for children under 5 was 18 times greater in sub-Saharan Africa than in Australia and New Zealand. The country with highest risk of mortality for children under 5 had 80 times greater risk than the country with the lowest risk.
- 4 out of 5 deaths in children under 5 occurred in sub-Saharan Africa and Southern Asia, but only 3 in 5 live births occurred in those regions.
- Sub-Saharan Africa accounted for 57% of global deaths under 5, but only accounted for 30% of global live births.
- Southern Asia accounted for a further 26% of global deaths under 5, but only accounted or 27% of global live births.
SDGs
The report states that, without “urgent action”, there is a risk of missing the SDG mortality targets of 25 or fewer under-5 deaths per 1,000 live births by 2030 (SDG 3.2.1) and 12 or fewer neonatal deaths per 1,000 live births by 2030 (SDG 3.2.2). At present, 59 countries are at risk of missing the SDG under-5 mortality target and 64 countries are at risk of missing the SDG neonatal mortality target.
- If we continue at the current rate, 35 million children under 5 are projected to die by 2030.
- Furthermore, 16 million children, adolescents, and youth between 5-24-years-old are projected to die by 2030.
- 9 million under-5 deaths could be prevented by accelerating progress to meet the SDG target in the countries at risk of missing the target.
Experts weigh in
UNICEF Executive Director Catherine Russell recognised that “behind these numbers lie the stories of midwives and skilled health personnel” who help deliver newborns, alongside the health workers and community health workers who provide vaccination and health and nutrition support.
“Through decades of commitment by individuals, communities, and nations to reach children with low-cost, quality, and effective health services, we‘ve shown that we have the knowledge and tools to save lives.”
However, Dr Tedros Adhanom Ghebreyesus, WHO Director General, emphasised that despite “welcome progress” there are still “millions of families” who “suffer the devastating heartbreak of losing a child”.
“Where a child is born should not dictate whether they live or die. It is critical to improve access to quality health services for every woman and child, including during in emergencies and in remote areas.”
Dr Juan Pablo Uribe, Global Director for Health, Nutrition, and Population, World Bank, and Director, Global Financing Facility for Women, Children, and Adolescents, commented that the report is an “important milestone”.
“But this is simply not enough. We need to accelerate progress with more investments, collaboration, and focus to end preventable child deaths and honour our global commitment. We owe it to all children to ensure they have access to the same health care and opportunities, regardless of where they are born.”
UN Under-Secretary-General for Economic and Social Affairs agreed that the data are “important to track progress”, but also emphasise a need for more work.
“They should also remind us that further efforts and investments are needed to reduce inequities and end preventable deaths among newborns, children, and youth worldwide.”
The full report can be accessed on the UN IGME page here. How might vaccination strategies factor into efforts to reduce the number of deaths and meet SDGs? For more on this, join us at the Congress in Washington this April or subscribe to our newsletters here.

by Charlotte Kilpatrick | Mar 6, 2024 | Technology |
In February 2024 Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X) announced that it is awarding GlyProVac $467,000 in support of efforts to develop a maternal vaccine targeting Escherichia coli (E. Coli), which is the bacterial species responsible for a “large portion” of neonatal sepsis infections. GlyProVac is a spin-out of the University of Southern Denmark in Odense that seeks to reduce the burden of bacterial infections and antimicrobial resistance worldwide.
Neonatal sepsis
Neonatal sepsis is a life-threatening response to bloodstream infections that occurs in newborns younger than 28 days old. Their immature immune systems mean they are “particularly susceptible” to infections. A recent study estimated that 2.5 million neonates or infants in the first month of lie die each year from sepsis; the greatest burden is suffered in low- and middle-income countries. With rapid disease progression, neonatal sepsis requires immediate treatment.
GPV02
The award will contribute to the development of GlyProVac’s maternal vaccine GPV02. GPV02 uses a selected bacterial protein, “naturally decorated” with small sugar molecules, to trigger an immune response. This contrasts to previous attempts to generate protein-based bacterial vaccines. GlyProVac uses BEMAP technology to ensure that the vaccine “correctly imitates” E. coli to prepare the immune system to recognise the bacterium when infection occurs.
The BEMAP technology is a “powerful sample enrichment method” based on mass spectrometry that allows comprehensive identification of any type of O-linked protein glycosylation. Through the technology the team has discovered that bacterial O-linked glycosylation is “much more extensive than previously thought and is especially important to Enterotoxigenic E. coli.
Dr Erin Duffy, CARB-X’s R&D Chief, commented that GlyProVac’s maternal vaccine could be important to prevent neonatal sepsis, the “leading cause of death among infants”.
“Because newborns at risk for neonatal sepsis are too young to be immunised, the vaccine would be administered to expectant mothers and target bacteria that cause neonatal bloodstream infections in babies. The vaccinated mothers would pass antibodies on to their babies in utero and through breastmilk after birth to strengthen their newborns’ immune systems, helping them ward off infections.”
Dr Anders Boysen, CEO and Co-founder of GlyProVac and Co-inventor of BEMAP is pleased that the award enables the “strengthening” of the team’s “scientific approach, strong team, and commitment to tackle the AMR crisis”.
“E. coli can cause a range of infections in humans, including urinary tract infections and neonatal sepsis in newborns, and poses a major challenge for public healthcare systems. We are grateful for the invaluable support, network, and resources provided by CARB-X, which will give us the opportunity to expand the potential of our E. coli vaccine to accelerate the fight against neonatal sepsis.”
To participate in important discussions about the role that vaccines can play in tackling the threat of AMR do get your tickets to join us at the workshops before the Congress in Washington this April and don’t forget to subscribe here.

by Charlotte Kilpatrick | Feb 14, 2024 | Infection |
In February 2024 a team from Dartmouth’s Geisel School of Medicine and Thayer School of Engineering announced that their research in Cell Reports offers “new insights” into how antibodies function in “combating” herpes simplex virus (HSV) infections. The study outlines how effector functions are “crucial” for protection by glycoprotein D (gD)-specific mAbs. It is hoped that this progress could facilitate treatments for neonatal herpes.
HSV
Herpes simplex virus infections are “common” and “typically” affect the skin and nervous system. They often lie dormant in the body without posing a serious health risk but can be “more dangerous for those with weak immune systems”. Neonatal herpes simplex virus infections are “particularly devastating”, with the potential for infections to spread to internal organs and the brain, causing “loss of life or long-term neurological disability”.
Although new treatment regimens have “improved outcomes”, the authors describe neonatal mortality following disseminated disease “unacceptably high”. Therefore, they identify a need for insights into “how Abs exert direct and indirect antiviral activities to protect against infection could aid in the design of both passive and active immunisation strategies”.
“Like other consequential early-life pathogens, most studies of HSV have focused on adult animal models. There is therefore a dearth of information on how Abs protect in the neonatal period.”
The team set out to investigate the mechanism(s) by which Abs that target gD mediate protection against nHSV-1 and nHSV-2 infections. Through a mouse model of nHSV infection they were able to demonstrate “distinct mechanisms” of Ab-mediated protection that differ between viral types.
Understanding the need
Dr David Leib, chair and professor of microbiology and immunology, commented that “despite three decades of trying, the scientific community has been unable to develop an effective vaccine against herpes”.
“I think the main issue has been that we haven’t fully understood what we need, in terms of antibodies and their specific functions, to protect against this disease.”
Dr Leib was surprised by the results, suggesting that the team discovered “something unexpected”.
“It’s not just the neutralising capability of antibodies, that is, their ability to bind directly to the virus and prevent it from entering the cell, that is important. Effector functions, which allow the antibodies to interface with other parts of the immune system, also play a critical role – one that has been largely overlooked in the past.”
The paper states that this research could contribute to mAb-based prevention and therapy as well as vaccine design. The authors emphasise the conclusion that “polyfunctional mAbs able to mediate both neutralisation and effector functions” will be the “best candidates” for therapeutic and prophylactic translation.
“Expanding the focus of vaccine research and development to include activities beyond viral neutralisation has the potential to accelerate the quest for interventions to reduce the global burden of HSV infection.”
How can we accelerate research and innovation to improve vaccine outcomes for neonates and older populations? To join the discussion at the Congress in Washington this April get your tickets here and don’t forget to subscribe!

by Charlotte Kilpatrick | Feb 9, 2024 | Global Health |
Researchers from Weill Cornell Medicine shared in February 2024 that an experimental mRNA vaccine against human cytomegalovirus (CMV) elicited “some of the most promising immune responses to date” of any CMV vaccine candidate. Their work, published in the Journal of Infectious Diseases, shows that the mRNA vaccine from Moderna could protect adults against CMV, which means it could also protect vertical transmission during pregnancy. They compared the vaccine with a “moderately successful” candidate to reveal that the mRNA vaccine was “better at preventing” the CMV virus from infecting epithelial cells and “more effective at triggering the immune system” to respond to CMV-infected cells.
A danger to newborns
The statement suggests that, although healthy adults are “largely asymptomatic”, one in every 200 newborns worldwide is infected with CMV during the mother’s pregnancy.
“The virus rarely causes serious illness in healthy adults, but it can cause birth defects and brain damage in newborns infected in utero and deadly infections in immune-compromised adults.”
Dr Sallie Permar from Weill Cornell Medicine explained that CMV is the “most common congenital infection worldwide”.
Comparing with earlier efforts
Sanofi and Novartis have previously developed vaccine candidate gB/MF59, which protected “about half of vaccinated individuals” from CMV infection in a Phase II trial led by the NIAID Vaccine Clinical Trials Network. However, after the trial concluded in 2013 it did not continue to Phase III. Dr Permar and her team were able to use the data and samples from the Phase II trial in adolescent girls as a benchmark.
Moderna’s vaccine candidate targets both the previously targeted glycoprotein B (gB) and a new target, a five-unit protein complex that allows the virus to infect the epithelial cells in the nose and mouth. In the latest study the researchers compared the immune responses of participants who had been vaccinated in the earlier trial with those vaccinated with the mRNA-based vaccine in a Phase I trial that ended in 2020.
The team believes that the addition of this second target enabled the mRNA vaccine to do a. ‘better job” of preventing infection. Dr Permar commented that the “newer vaccine has the potential to be more effective” because “some of the functional immune responses it elicits are higher in magnitude”.
“An ongoing clinical trial will confirm if those differences lead to greater protection against CMV infection.”
The vaccine has progressed to the “first ever” Phase III clinical study for a CMV vaccine candidate.
“After more than 50 years of research, we are closer than ever to having a licensed CMV vaccine. The new mRNA platform has a lot of potential.”
Dr Permar’s team has also developed a preclinical model to test the protection afforded by similar vaccines against foetal CMV transmission during pregnancy.
For more on vaccine development and progress in infection control, don’t forget to get your tickets to join us at the Congress in Washington this April or subscribe to our newsletters here.

by Charlotte Kilpatrick | Feb 7, 2024 | Global Health |
In February 2024 Karolinska Institutet announced that the results of a study in JAMA reveal that COVID-19 vaccination during pregnancy did not increase risk for babies, and “for some serious neonatal complications” lowered risks. The paper emphasises the importance of understanding neonatal adverse events after COVID-19 vaccination during pregnancy to “help address concerns about vaccine safety”. Therefore, a collaboration between teams in Sweden and Norway investigated the effects of any mRNA vaccine against COVID-19 on infants whose mothers received it.
Assessing vaccination risk
The authors recognise that vaccination during pregnancy has been associated with “reduced rates of COVID-19 in pregnant individuals and their newborn infants”, which has led different authorities to recommend vaccination during pregnancy. However, the balance between benefits and risks of COVID-19 vaccination during pregnancy “may be more nuanced” for newborn infants than the vaccinated person. Indeed, “concerns about potential adverse foetal and neonatal effects” have influenced vaccination rates in pregnant individuals.
“Vaccination has not been associated with adverse pregnancy outcomes or higher admission rates to neonatal care, but information on neonatal outcomes is limited.”
This study, a large population-based cohort study, sought to perform a “comprehensive assessment of neonatal safety” for mRNA COVID-19 vaccines. To address “unease” about potential neonatal brain lesions associated with COVID-19 vaccination, priority was given to exploring neonatal cerebrovascular outcomes.
What did the study find?
Of 196,470 newborn infants included, 94,303 (48%) were exposed to COVID-19 vaccination during pregnancy. There was “no evidence” of increases in neonatal adverse events for these infants. In fact, exposure to COVID-19 vaccination during pregnancy was associated with reduced rates of nontraumatic intracranial haemorrhage, hypoxic-ischemic encephalopathy, and neonatal mortality. Although the observed reduction in risk could reflect “residual confounding”, the authors assume that the results will offer “reassurance” to public health authorities, clinicians, pregnant people, and their families.
“The results of the current study were overall reassuring, with no indication of increased risks for cerebrovascular conditions in newborn infants after COVID-19 vaccination in pregnancy with mRNA vaccines.”
Furthermore, previous research was corroborated by the present data, which do not suggest an increased risk of hypoxic-ischemic encephalopathy. Other concerns include pericarditis or myocarditis as a potential adverse effect of mRNA COVID-19 vaccination in young people, particularly boys.
“Reassuringly and irrespective of mRNA vaccine manufacturer or trimester of administration, there were no cases neonatal myocarditis among infants after maternal vaccination during pregnancy.”
There were also “no indications of an increased risk for other inflammatory neonatal diseases”. The authors highlight that the prepandemic neonatal mortality in 2019 in Sweden and Norway was 1.3 and 1.2/1000, respectively. A higher neonatal mortality (2.0 and 1.6/1000) was observed in pregnant individuals who were not vaccinated during pregnancy. This “may reflect a higher likelihood” of “unfavourable risk” factors associated with “lower socioeconomic status and poorer living conditions than the general pregnant population”.
“It is unlikely that mRNA COVID-19 vaccination during pregnancy directly reduces neonatal mortality, although others have suggested that such a protective effect is biologically plausible.”
Professor of Paediatrics and Neonatology at the Department of Clinical Science, Intervention and Technology, Karolinska Institutet, Mikael Norman, emphasises that previous research revealed that “the vaccine does not cross the placenta” and “cannot be found in umbilical cord blood”. He recognised that “COVID-19 is still present” and is likely to be around for “a long time”.
“It is therefore very important for the one hundred thousand women who become pregnant every year in Sweden, and the 130 million in the world, to know that vaccination with mRNA-vaccines against COVID-19 is safe for their babies. We found no increased risks; if anything, infants vaccinated women had lower risks for some severe outcomes.”
We’re looking forward to exploring the risks, benefits, and challenges of COVID-19 vaccines at the Congress in Washington this April. To join us, get your tickets today and don’t forget to subscribe for more insights!

by Charlotte Kilpatrick | Jan 29, 2024 | Global Health |
In January 2024 the University of California Los Angeles (UCLA) shared that results from a study in Nature Communications reveal that infants born full to term to mothers who had been infected with COVID-19 during pregnancy had a three times greater risk of experiencing respiratory distress (RD). The risk was “significantly lower” if mothers who were infected during pregnancy had been vaccinated.
Respiratory distress
The study authors note that SARS-CoV-2 infection during pregnancy has been associated with “adverse maternal and neonatal outcomes”. Although estimates of vertical transmission from mother to child are “low”, they report “growing concern for long-term neonatal consequences”. Furthermore, research has identified cases of infant respiratory distress (RD) among SARS-CoV-2 exposed uninfected (SEU) term neonates.
Previous studies have attributed cases of RD among these SEU neonates to “maternal hypoxia and multiorgan failure”, which increases the risk of premature delivery. However, for infants born at term, the increased cases of RD remain unexplained.
“Our prior analysis suggests that prenatal exposure to SARS-CoV-2 may activate an inflammatory cascade in the newborn airway leading to ciliary beat dysregulation of the airway epithelium.”
Furthermore, inflammasome associated proteins (IL-18, IL-1B, and CASP1) have been identified in SEU term infants with RD, but the “exact mechanism of this pathway has yet to be elucidated”. The study considers that maternal immunisation against COVID-19 has been demonstrated “highly protective” against maternal mortality and morbidity, possibly preventing COVID-19 hospitalisations in infants under 6 months of age.
The study
The research explores the “complex relationship between maternal SARS-CoV-2 infection during pregnancy and infant RD”. The team considered 3 objectives:
- Characterise the clinical features of RD among SEU infants
- Construct a multivariable statistical model to assess the effect of maternal vaccination on infant RD
- Conduct a proteomic ingenuity pathway analysis to identify proteins differently expressed in SEU infants and their biological functions
“Our study highlights the importance of public health interventions and vaccination efforts that target pregnant individuals due to the potential for lasting effects on the health of both the mother and the infant.”
221 mothers were enrolled in the study, of which 151 (68%) were unvaccinated before their infection. Of these, 23 women (16%) experienced severe or critical disease, compared with 3 (4%) of vaccinated mothers. 34 (17%) of the 199 exposed infants that were followed had respiratory distress, a “high frequency” compared to the general population. The team found that of the 34 infants with respiratory distress, only 5 (16%) were born to mothers who had been vaccinated prior to infection, compared to 63 (41%) without the breathing disorder. This suggests that vaccination had a “protective effect”.
Senior author Dr Karin Nielsen, professor of paediatrics at the David Geffen School of Medicine at UCLA, commented that “unusually high rates” of respiratory distress were observed in babies born to mothers who had COVID-19 during pregnancy.
“The mothers had not been vaccinated prior to acquiring COVID, indicating that vaccination protects against this complication.”
How good is the study?
UCLA and the authors recognise the limitations of the study, including a possible skew towards “more severe” illness compared to the general population. On the other hand, because of a “high level of specialised services and medical equipment”, the people in the study may have “higher rates of survival” than other regions.
“While our findings suggest that the magnitude of the effect of in utero COVID-19 exposure on neonatal RD is high, the absolute number of cases remains small and limits the power of our study. Given our small sample size, our results should be interpreted with caution.”
Do you find the study provides a useful contribution to the discourse around maternal vaccination and protection of neonates? If COVID-19 vaccination is a subject of interest to you, do join us in Washington for the Congress in April, or subscribe to weekly updates on all things vaccine!