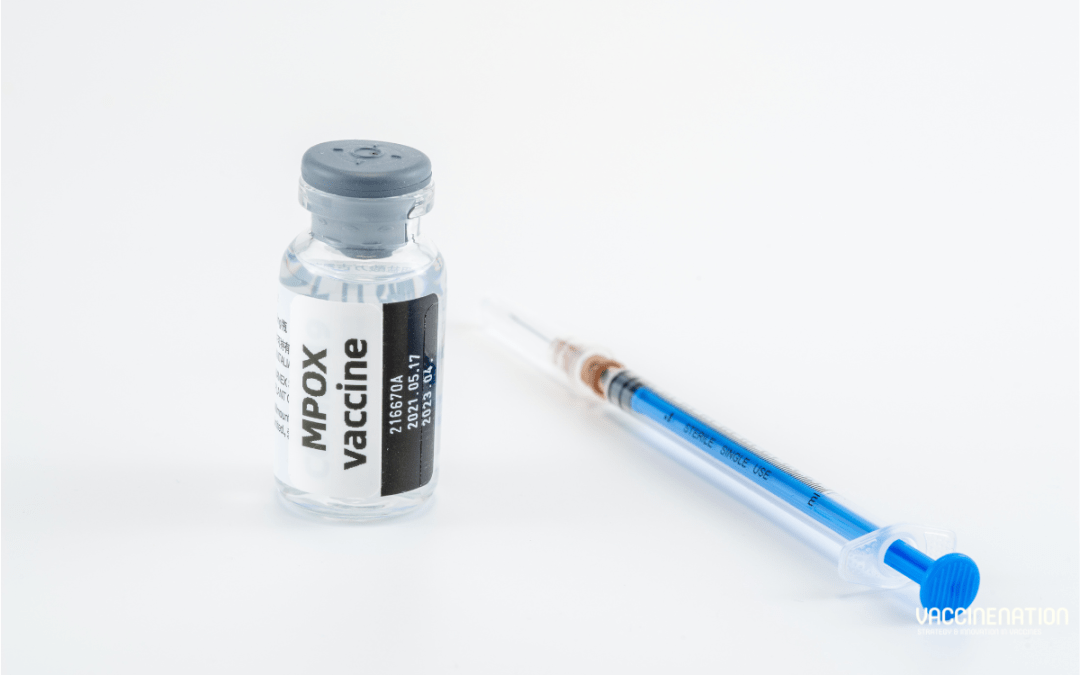by Charlotte Kilpatrick | Oct 23, 2024 | Global Health |
A study in The Lancet Global Health sought to provide counterfactual scenarios to evaluate the short-term effects of different vaccination strategies on mpox cases and deaths in the Democratic Republic of the Congo (DRC). The researchers used a dynamic transmission model to simulate mpox transmission, stratified by age and province; this was used to assess potential vaccination strategies and their effects on deaths and cases in an epidemic year. The results indicate that vaccinating children aged 15 years or younger, or younger than 5 years, in endemic regions, would be the “most efficient use of vaccines” when resources are limited.
Mpox in DRC
Mpox was first identified in the Democratic Republic of the Congo (DRC) in 1970; it is a zoonotic infectious disease caused by the monkeypox virus (MPXV), which is endemic in “numerous regions” of west and central Africa. MPXV has two clades:
- Clade I is endemic in central Africa with an estimated case fatality rate of up to 10% and mainly affecting children. It is divided into two subclades, Ia and Ib.
- Clade II was historically found in west Africa, with an estimated case fatality rate of up to 1%-3%. It is also divided into two subclades, IIa and IIb. Clade IIb was responsible for the global mpox epidemic in 2022.
The authors state that, until 2022, MPXV was not associated with large outbreaks; most cases were related directly to sylvatic transmission from animals to humans via hunting, wild game preparation, and consumption. Increases in human-to-human transmission were identified in 2017.
The researchers suggest that the low likelihood of transmission in the early decades after the virus’ discovery could be related to smallpox eradication programmes, which offered cross-immunity via vaccination against a related orthopoxvirus. Indeed, since the cessation of the smallpox vaccination programme in the DRC, there has been a “concurrent increase in mpox cases and outbreak frequency”. There is an ongoing, “unprecedentedly large” outbreak of clade I mpox in the DRC, with more than 14,000 reported suspected cases by the end of 2023 and a 4.6% case fatality rate. Over 70% of the deaths are in children younger than 15 years.
Genetic analyses of clade Ia MPXV genomes indicate that in this outbreak, multiple, independent zoonotic introductions into the human population have occurred from one or more reservoir species. An increasing burden of clade Ib MPXV infections have been identified in eastern DRC with evidence of “sustained” human-to-human transmission and many cases in women aged 15-29 years, but clade Ia infections continue to comprise most mpox cases in the DRC.
The study
Bavarian Nordic’s modified vaccinia Ankara vaccine (JYNNEOS) is protective against mpox. It was approved by the US FDA in 2019 but was not widely used against mpox until the 2022 outbreak, when it was “quickly mobilised to vaccinate people at high risk of infection in the USA and Europe”. Despite its high efficacy at two doses, it is “largely unavailable” outside the USA and Europe.
The authors aimed to inform policy and decision makers on the “potential benefits of, and resources needed,” for mpox vaccination campaigns in the DRC. They used an approach based on models from operations research and decision science to offer a robust analysis of policy choices “even in the context of incomplete and uncertain data”. The study uses mathematical modelling to simulate the spread of mpox in the DRC during 2023.
Without vaccination, the model predicted 14,700 cases of mpox and 700 deaths from mpox in the DRC over 365 days, consistent with reported estimates. Almost 50% of the cases and deaths came from the province of Equateur. Cases were evenly split between the three age groups: 34% in children under 5 years, 32% in children aged 5-15 years, and 34% in people older than 15 years. However, deaths were “predominantly” seen in children younger than 5 years (51%).
Vaccinating 80% of children younger than 5 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 10,500 cases and 400 deaths. Vaccinating in endemic provinces increased cases to 10,700 and deaths remained the same. The numbers of vaccine doses needed for the strategies were 41.4 million (all provinces), 33.8 million (provinces with a history of mpox), and 13.2 million (endemic provinces only).
Vaccinating 80% of children younger than 15 years in all provinces or provinces with a history of mpox cases decreased the outbreak to 6,400 cases and 200 deaths. Vaccinating in endemic provinces increased cases to 6,800 and deaths remained the same. The numbers of vaccine doses required for these strategies were 81.6 million (all provinces), 67.1 million (provinces with a history of mpox), and 26.6 million (endemic provinces only).
Vaccinating 80% of all ages in all provinces or only non-endemic provinces with a history of cases decreased the case burden to 1,400 cases and 100 deaths, and 2,000 cases and 100 deaths when vaccinating in provinces endemic for mpox. The numbers of doses required for these strategies were 170.8 million (all provinces), 142.0 million (provinces with a history of mpox), and 56.8 million (endemic provinces only).
Managing resources
The paper finds that vaccinating all ages leads to the “largest impact on magnitude of cases and deaths”, but that vaccinating only children aged 15 years or younger provides “nearly the same effect with fewer vaccine doses required”. Although vaccinating only children younger than 5 years showed a “drop-off” in averted cases and deaths, it provides the most efficiency.
“This analysis shows the effectiveness of focussing an mpox vaccination campaign specifically in the provinces endemic for mpox in the DRC. This targeted strategy prevents nearly as many cases and deaths as broader approaches but uses fewer vaccine doses and thus would be less costly to implement.”
Alexandra Savinkina, fourth year PhD student in the Yale School of Public Health (YSPH) Department of Epidemiology (Microbial Diseases), commented that this study could influence vaccination policy.
“My hope is that it could help inform policy for vaccination in the country and potentially the region and move the needle forward on getting vaccines to the people who need them most in the DRC.”
Savinkina hopes that “we can learn from the global mpox outbreak that we can’t ignore disease in other places”.
“If the resources to help people exist, I think we should be using them, whether in the U.S. or in Africa.”
Dr Gregg Gonsalves, associate professor of epidemiology at YSPH, acknowledged barriers to access.
“We take it for granted that we can get a vaccination for COVID or a flu shot at our local CVS, but the infrastructure to deliver vaccines in DRC is far less robust.”
For more vaccine research updates, subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 16, 2024 | Global Health |
In October 2024, parliamentarians from across the world met at the UNITE Global Summit in Berlin to sign a statement in support of the WHO Pandemic Agreement. The statement was described by WHO as a “significant commitment” to “strengthen pandemic preparedness, response, and equitable access to health”. The Summit was held in collaboration with the World Health Summit (WHS), drawing global parliamentarians, civil society leaders, and health experts to “translate discussions into actionable policy priorities”.
Building trust for a healthier world
The World Health Summit 2024 was held under the theme “building trust for a healthier world”, focussing on inspiring “innovative solutions for better health and well-being for all”. The UNITE Global Summit agenda reflected key topics highlighted during WHS, divided into four pillars:
- Human rights and equitable access to health
- Global health architecture and security
- Strengthening of healthcare systems
- Sustainable financing for health
Support for the Pandemic Agreement
The Pandemic Agreement is under negotiation by WHO Member States as an attempt to address the “gaps” exposed by the COVID-19 pandemic and the threats presented by mpox and other disease outbreaks. It also seeks to “strengthen global collaboration pandemic prevention, preparedness, and response”.
A critical moment during the summit was the signing of the Global Parliamentary Statement in Support of the Pandemic Agreement, which demonstrated the importance of parliamentarians in “ensuring global health security and safeguarding populations against future pandemics”. The statement emphasised their commitment to ensuring that “all countries, regardless of resources, have access to the tools, capacities, resources, and healthcare required” during a pandemic.
Dr Ricardo Baptista Leite, President of UNITE, commented that parliamentarians are the “voice of the people” and carry a “crucial responsibility in safeguarding public health”.
“The WHO Pandemic Agreement represents a historic opportunity to prevent pandemics and strengthen our global preparedness and response capabilities. By signing this statement, we are not only showing our support for the agreement but also pledging to ensure that its principles of equity, solidarity, and global cooperation are fully realised in every nation.”
WHO Director-General Dr Tedros Adhanom Ghebreyesus welcomed the support for the “once-in-a-generation opportunity to build a stronger, fairer, and more prepared global health system”.
“By signing this statement, parliamentarians from around the world are showing their commitment to protecting lives from future pandemics and ensuring equitable access to vaccines, treatments, diagnostics, and other health tools for every country, particularly those with fewer resources.”
Key commitments
The Global Parliamentary Statement emphasises four “key commitments”:
- Equity at the core – ensuring equitable access to pandemic-related health tools based on public health need for everyone, especially countries with fewer resources
- Global solidarity – strengthening international cooperation to build resilient health systems that can prevent and respond to pandemics
- Legislative action – advocating for the ratification and implementation of the Pandemic Agreement within national legislatures, as appropriate
- Combating misinformation – providing communities with evidence-based health information to counter the spread of harmful misinformation
Dr Baptista Leite reflected that “the challenges we face today demand a global response”.
“No single country can prevent or combat pandemics alone. The WHO Pandemic Agreement is an essential step forward in ensuring that every nation has the tools, resources, and capabilities to respond to future health threats.”
More work to be done
WHO states that the Pandemic Agreement “needs to continue to garner broad international support”. Within the latest commitment, parliamentarians are to work with WHO and other international organisations to ensure the Pandemic Agreement is implemented in a way that “benefits all countries”, particularly those with “limited resources”.
“The statement signed in Berlin is expected to act as a catalyst for global parliamentary action, fostering collaboration and solidarity among nations.”
Join us at the Congress in Barcelona this month for a keynote discussion on “strengthening global preparedness and building resilient health systems” with experts from the field, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 14, 2024 | Global Health |
Phase II trials of a “promising” human vaccine candidate against Rift Valley fever are beginning in Kenya with CEPI support. CEPI reported in October 2024 that this is the “most advanced stage of testing” for a human Rift Valley fever vaccine in an “outbreak-prone area”. Scientists at the University of Oxford and the Kenya Medical Research Institute (KEMRI)-Wellcome Trust Research Programme are leading the trial with $3.7 million funding from CEPI.
Rift Valley fever
First identified in Kenya’s Rift Valley, Rift Valley fever is usually found in people after direct contact with infected animals or bites from infected mosquitoes. Most infected people experience “mild disease”, but a small proportion develop the “severe haemorrhagic form”, with a risk of blindness, convulsions, encephalitis, and bleeding. In these cases, mortality rates can reach 50%.
Rift Valley fever has been detected across “much of Africa” and in the Middle East. It is mosquito-borne, which makes it “climate sensitive”. There is therefore a risk of outbreaks spreading to new areas or increasing in frequency or size. There are Rift Valley fever vaccines for animals, but no currently available or licensed vaccines for human use; the disease is a priority disease for R&D for WHO and CEPI.
ChAdOx1 RVF in trial
The vaccine, known as ChAdOx1 RVF, is based on the University of Oxford’s ChAdOx1 vaccine platform. It has shown positive results in healthy adults in the UK, meeting “many of the optimal product characteristics” of a WHO target product profile. It is one of three Rift Valley fever candidates in CEPI’s portfolio. 240 healthy adults will participate in the research, following local trial approvals.
Funding for the trial comes under CEPI’s strategic partnership with the University to accelerate the development of globally accessible vaccines against outbreak pathogens. Both organisations are “committed to enabling access to any vaccine outputs developed through this partnership”, including developing a target product profile suitable for low- and middle-income countries (LMICs), assessing the need for technology transfer, and priority supply to LMICs at an affordable price.
Professor George Warimwe, Principal Investigator of the trail and Deputy Executive Director of the KEMRI-Wellcome Trust Research programme, reflected that “nearly 100 years” after the disease was identified, there are “still no approved vaccines or treatments”.
“This vaccine trial brings us closer to addressing the rising frequency of outbreaks.”
Dr Richard Hatchett, CEO of CEPI, commented that Rift Valley fever “disproportionately affects the lives and livelihoods of vulnerable pastoral communities”, causing human fatalities and livestock losses.
“Investing in the promising human ChAdOx1 RVF vaccine diversifies CEPI’s portfolio and gives us a greater chance at protecting vulnerable populations against this worrisome threat that may become more prevalent with climate change.”
Director General of Africa CDC, H.E. Dr Jean Kaseya, agreed that the disease “leads to livestock losses and human fatalities, thus impoverishing communities who largely depend on livestock for their livelihood”.
“The launch of a Phase II clinical trial of a Rift Valley fever vaccine candidate in an endemic country is a crucial milestone in our efforts to control this disease. Africa CDC is proud to support this initiative that not only prioritises the health of our people but also demonstrates the continent’s growing leadership in advancing clinical research.”
Dr Kaseya stated that the ChAdOx1 RVF vaccine “offers hope to vulnerable populations” who are “disproportionately affected by the growing impact of climate change”.
Join us at the Congress in Barcelona later this month to explore efforts to address the growing challenges of climate change and infectious disease with vaccine development and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 10, 2024 | Global Health |
In October 2024, WHO announced an agreement with the International Monetary Fund (IMF) and the World Bank Group (WBG) on “broad principles for cooperation on pandemic preparedness”. The cooperation is intended to enable scaling up of support to countries to prevent, detect, and respond to public health threats through the IMF’s Resilience and Sustainably Trust (RST), WBG’s financial and technical support, and WHO’s technical expertise and in-country capabilities. The organisations will “leverage their experience to enhance pandemic preparedness”, working on the “synergies and complementarity” of each institution’s in-country analysis and operations.
Principles of coordination
Under the Broad Principles of Coordination:
- WHO and the WBG will continue to lead on health-related development policies and, with other multilateral development banks and The Pandemic Fund, on specific project investments for pandemic preparedness. RST financing will not be earmarked for specific projects.
- Pandemic preparedness policy reform measures supported by RSF arrangements will be informed by existing data, analytics, and operational engagement of WHO, the WBG, and country authorities.
- Pandemic preparedness reforms will build on each institution’s area of expertise. RSF programmes will focus on macro-critical policy reforms within the IMF’s expertise and complement the work carried out by the WBG and WHO to maximise both the financial resources and technical expertise available to countries. RSF Reform measures can include policy actions aimed at enhancing the readiness of finance and health systems to respond effectively to future health emergencies.
The cooperation will enable all three institutions to better serve countries’ efforts on pandemic preparedness.
Working for a safer world
Kristalina Georgieva, Managing Director of the IMF commented that the “stepped-up collaboration” will help the organisations to “complement and leverage each other’s expertise” to support members’ pandemic preparedness and resilience efforts.
“The IMF’s Resilience and Sustainability Trust allows eligible member countries to access affordable, long-term financing to address structural challenges that threaten their macroeconomic stability.”
WHO Director-General Dr Tedros Adhanom Ghebreyesus reflected on the need for “new sources of financing to bolster health systems”, making them “more able to prevent and detect” health threats and to “respond and withstand them when they strike”.
“WHO is proud to be working with the IMF and the World Bank to unlock financing from the Resilience and Sustainability Trust, and support countries to put it to work for a safe world.”
World Bank Group President Ajay Banga suggested that the “deepened collaboration” will focus efforts to help countries prepare for and respond to health threats.
“We must aggressively be planning and preparing for the next global health crisis, so that when the battle comes – and we know it will – we will have the health workforce that can be rapidly deployed in the face of a crisis, laboratories that can quickly ramp up testing, and surge capacity that can be called upon to respond.”
For insights into pandemic preparedness initiatives at the Congress in Barcelona this month get your tickets here, and don’t forget to subscribe to our weekly newsletters here!

by Charlotte Kilpatrick | Oct 8, 2024 | Infection |
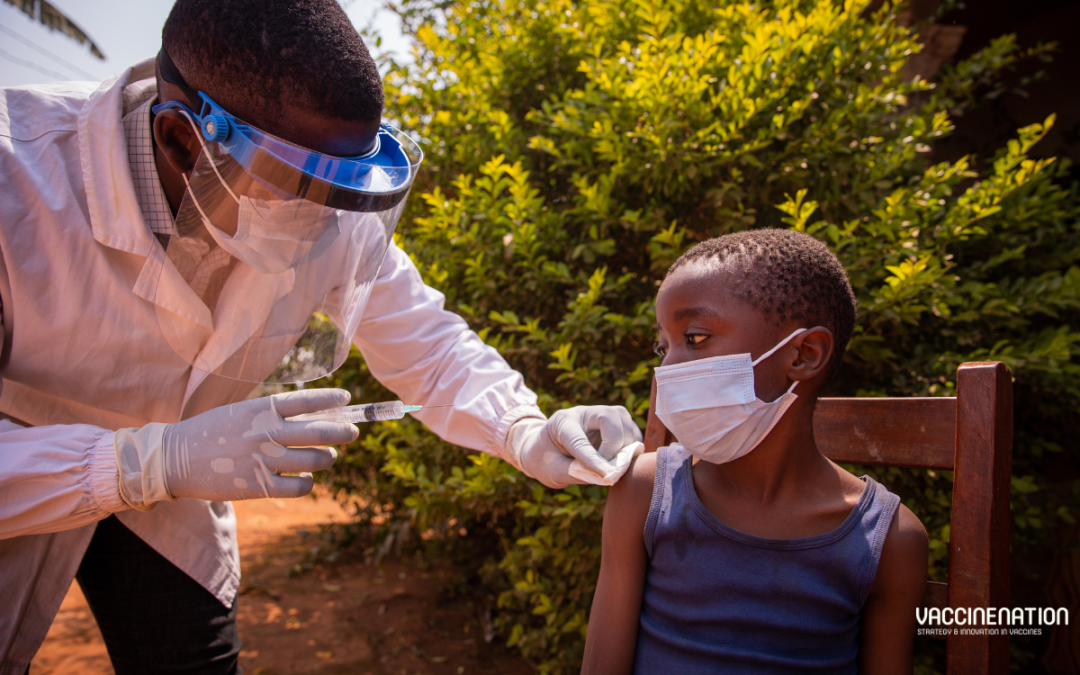
A recent WHO situation report on the mpox PHEIC reveals that 14 countries on the continent have reported mpox cases in the last six weeks and are considered to have “active” outbreaks. The most affected country in 2024 is still the Democratic Republic of the Congo (DRC), which recently announced the start of an mpox vaccination campaign. The update presents reported confirmed mpox cases and deaths as well as reported suspected mpox cases, as defined by the countries that have reported them.
Risk levels
The report presents the mpox risk of geographical spread and potential impact on health in various regions:
- Eastern Democratic Republic of the Congo and neighbouring countries: high
- Areas of the Democratic Republic of the Congo where mpox is endemic: high
- Nigeria and other areas of West, Central, and East Africa where mpox is endemic: moderate
- All other countries in Africa and around the world: moderate
However, the report notes that individual country or regional bloc assessments may vary, and the risk could be assessed as low. Individual-level risk is “largely dependent on individual factors” like exposure risk and immune status.
Cases
By 29th September, 14 countries on the continent have reported mpox cases in the last six weeks. The most affected country in 2024 is the Democratic Republic of the Congo (DRC) with 5,610 confirmed cases and 25 deaths, followed by Burundi, with 853 confirmed cases and no deaths, and Nigeria, with 78 confirmed cases and no deaths. Although the epidemic curve of confirmed cases by country suggests a decline in reported cases in DRC, this trend “should be interpreted with caution” amid “reports of limited testing and stockout of testing supplies”.
Focus on North Kivu
As of 28th September 2024, the province of North Kivu in DRC had reported 323 confirmed mpox cases, including two confirmed deaths. After initial detection in May 2024 genomic sequencing analysis revealed clade Ib monkeypox virus (MPXV). There are 34 health zones in North Kivu; 14 have reported confirmed mpox cases, The most affected zones are Goma, Karisimbi, and Nyiragongo. 1,108 suspected mpox cases in North Kivu have been tested with a test positivity of 29%. The number of confirmed cases in the province continues to increase.
Among confirmed cases, 117 (36%) are individuals living in camps for Internally Displaced People (IDP), in the three most affected health zones. 13 IDP camps have reported at least one mpox case; the most affected are Munigi, Mugunga, Rusayo, and Kanyaruchinya. 8 out of 14 IDP camps (57%) have reported only one case, which indicates sporadic introduction, likely from outside the camp. Transmission may be sustained in the other six, which show more cases over time.
In North Kivu, around 50% of confirmed mpox cases are among adults and 54% of total cases are male. However, in IDP camps, approximately 75% of mpox cases are among children up to 17 years old and are evenly distributed between males and females. WHO believes that mpox transmission in North Kivu is “exclusively” human-to-human, mainly at the community level. 117 out of 323 (36%) cases have a known epidemiological link. The mode of transmission for 109 cases (34%) is reported to be sexual contact; among these cases, 57 (52%) are reported among female sex workers.
216 out of 323 (67%) cases have recovered from the disease; 109 are in isolation, 33 in the household, and 76 in healthcare facilities. 19 (9%) cases have presented complications and two have died in hospital.
Clade Ib detected in India
On 1st October 2024, the Ministry of Health and Family Welfare of India notified WHO of the first mpox case due to MPXV clade Ib. The National Focal Point reports that the case is an adult male, Indian national, with a recent history of international travel to the United Arab Emirates (UAE). The patient developed mpox symptoms on 8th September in UAE before arriving in India on 13th September. On 16th September he was admitted to a public hospital.
On 19th September, samples were tested at the National Institute of Virology (NIV) in Pune, confirming MPXV infection. The patient recovered without complications and was discharged on 30th September 2024. The health authorities in UAE are conducting a detailed case and contacts investigation to finalise the “comprehensive verification and validation process as per IHR procedures”.
This is the second reported case of this clade MPXV infection in the WHO South-East Asia Region (SEAR) and the third reported case of clade Ib MPXV infection outside the African Region. However, the first clade Ib infection in SEAR, reported in Thailand, and the clade Ib infection in Sweden, had a recent history of travel to affected countries in Africa, which this case did not.
Vaccine updates
WHO indicates that it is finalising the issuance of prequalification age extension of MVA-BN for persons 12-17 years old after European Medicines Agency (EMA) authorisation. It is providing “strategic and technical support” to the African Vaccine Regulatory Forum (AVAREF) and issuance of emergency use authorisation for MVA-BN to national regulatory authorities. In collaboration with AFRO, DRC, Ghana, Nigeria, Rwanda, and Tanzania, WHO is harmonising the cohort safety event monitoring protocol following mpox vaccination and ensuring global data collection.
With receipt of 265,000 doses of MVA-BN, DRC has begun a vaccination campaign in North Kivu with the intention of expanding to 11 of the most affected health zones across various provinces.
Join us at the Congress in Washington in April next year to reflect on the global response to this outbreak and hear updates on continued mpox vaccine development, and don’t forget to subscribe to our weekly newsletters for more insights.

by Charlotte Kilpatrick | Oct 7, 2024 | Global Health |
WHO Africa reported in October 2024 that the Democratic Republic of the Congo (DRC) has started a vaccination campaign as part of outbreak control efforts against mpox. The vaccination drive has launched in the eastern North Kivu province and will prioritise health workers and frontline responders, contacts of confirmed cases, contacts of those contacts, and other at-risk groups. It will later be implemented in 11 of the most affected health zones in Equateur, North Kivu, Sankiri, South Kivu, Sud-Ubangi, and Tshopo provinces.
Addressing the emergency
The Democratic Republic of the Congo (DRC) has reported more than 30,000 suspected and laboratory-confirmed cases and 990 deaths since the start of 2024. These numbers account for 90% of the cases reported from 15 countries in the African region this year. WHO recommends that vaccination should form part of a “comprehensive response” involving enhanced surveillance, community engagement, and case management. It is working with partners and the national authorities to “scale up and reinforce all the key control measures”.
In preparation for the mpox vaccination campaign, WHO has supported national health authorities in training health workers, enhancing vaccine delivery systems and infrastructure, and community engagement. There are also efforts to “reinforce measures to identify and address” vaccine misinformation and disinformation, responding with increased access to accurate information.
Delivering doses to affected areas
Noting that mpox vaccines are “currently in short supply, especially in Africa”, WHO is encouraging global collaboration to get doses to the people who need them most. In September, WHO prequalified MVA-BN for mpox, which is “expected to facilitate timely and increased access”. It is also working with partners like Gavi and UNICEF to establish a distribution mechanism for donated doses and direct procurements. DRC has received 265,000 doses of MVA-BN, donated by the European Commission’s Health Emergency Preparedness and Response Authority, Gavi, and the United States Government.
WHO Regional Director for Africa, Dr Matshidiso Moeti, expressed gratitude to these partners for their donations.
“As we rally efforts to stop the mpox outbreak, the rollout of the vaccine marks an important step in limiting the spread of the virus and ensuring the safety of families and communities.”
Dr Moeti commented that WHO is “working closely with the national authorities to effectively deliver the vaccines to those who need them most”. Africa CDC also recognised the collaborative effort, which “underscores the collective global commitment” to controlling the outbreak in Africa. H.E. Dr Jean Kaseya, Africa CDC Director General, commended the DRC’s “swift action” in launching the campaign, which “showcases the strength of its public health leadership”.
“By prioritising vulnerable populations, including frontline health workers and those most at risk, the country is taking critical steps to contain the outbreak. Africa CDC remains committed to working closely with the DRC to ensure vaccines reach those who need them the most, while also working to strengthen health systems to prevent future outbreaks. Our top priority is to secure safe and effective vaccines for children in the next phase of vaccination.”
Mpox vaccination will be a key area of high-level discussions at the Congress in Washington next April, including on a keynote panel that will consider the “role of vaccines in a changing world”. Get your tickets to join us for these conversations, and don’t forget to subscribe to weekly vaccine updates here.

by Charlotte Kilpatrick | Oct 7, 2024 | Global Health |
A week after the declaration of a Marburg outbreak in Rwanda in September 2024, Sabin Vaccine Institute announced that it is providing doses of its investigational Marburg vaccine to support the outbreak response. An initial shipment of approximately 700 doses will be used in a trial involving frontline workers, including healthcare professionals, who have been the “hardest hit” by this outbreak. Sabin and the Rwanda Biomedical Centre have entered a clinical trial agreement for a Phase II rapid response open label study, which will be conducted at six trial sites in Rwanda. Sabin also plans to supply additional vaccines, pending a request from Rwandan officials and authorisation from BARDA.
Responding to the outbreak
The outbreak was declared on 27th September 2024, and by 6th October it had caused 12 deaths. Many cases are among health workers in two facilities in Kigali, but there are more cases spread across other districts. Sabin has been “working directly” with Rwandan officials and partners to support the response. There are no licensed vaccines or treatments for Marburg, but Sabin’s single-dose vaccine is in Phase II trials in Uganda and Kenya, with no safety concerns reported to date. The vaccine is based on the ChAd3 platform and results from Phase I clinical trials and nonclinical studies suggest that it is safe and elicits “rapid” and “robust” immune responses.
Commenting on the support from Sabin, Rwanda’s Minister of Health Dr Sabin Nsanzimana reflected that “in emergency situations, the success of clinical trials relies on quick, strategic global partnerships” that combine “expertise, resources, and innovation”.
“Today, a week after this Marburg outbreak was first confirmed, we are receiving doses of the Sabin Vaccine Institute’s Marburg vaccine candidate to protect our health workers and other high-risk groups, and also advance scientific tools which will ensure this virus can be effectively controlled now and in the future.”
Lightning speed
Sabin’s Chief Executive Officer Amy Finan described the team’s “lightning speed” in responding to the Rwandan government’s request for assistance, preparing shipments, finalising protocols, and securing regulatory and legal approvals.
“This swift emergency response demonstrates that a dedicated, collaborative group of individuals and organisations can achieve remarkable results when united by a common cause: to contain a lethal disease outbreak and prevent further loss of life.”
ReiThera, Sabin’s manufacturing partner, has produced the drug substance and filled and finished doses for shipment. CEO Stefano Colloca stated that the ReiThera team believes in the “transformative power of global collaboration to advance science and create lasting impact”.
“Our partnership with Sabin highlights our shared commitment to developing a life-saving vaccine against Marburg disease with a mutual goal: to save lives and ensure that even the most vulnerable communities around the world have access to vital and equitable protection.”
Sabin’s vaccine progress
Sabin plans to launch a Phase II trial of the Marburg candidate in the United States next year, as it looks forward to interim results from the trial in Uganda and Kenya. The development programme is supported by BARDA, which has committed $235 million for advancing vaccine research and development against Sudan ebolavirus and Marburg virus diseases.
To join discussions about safety and effectiveness evaluations of vaccines deployed in emergency situations, get your tickets to the Congress in Barcelona this month. Don’t forget to subscribe to our weekly newsletters for vaccine updates.

by Charlotte Kilpatrick | Oct 3, 2024 | Global Health |
WHO announced in October 2024 that it is launching the Global Strategic Preparedness, Readiness, and Response Plan (SPRP) to tackle dengue and other Aedes-borne arboviruses. The Plan is intended to reduce the burden of disease, suffering, and deaths from dengue and other Aedes-borne arboviral diseases, like Zika and chikungunya, by “fostering a global coordinated response”. It presents priority actions to control transmission and offers recommendations to affected countries across various sectors. With five “key components”, the Plan is to be implemented over one year until September 2025, demanding US$ 55 million.
“The SPRP is a call to action for all stakeholders – from government agencies and health-care providers to communities and individuals – to join forces in the fight against dengue and other Aedes-borne arboviruses, through innovation, new technologies, and improved vector control strategies.”
Turning the tide
In the foreword by WHO Director-General Dr Tedros Adhanom Ghebreyesus we learn that dengue has “afflicted humanity for centuries, and possibly longer”; the first report of a clinically compatible case is recorded in a Chinese medical encyclopaedia in 992. From a much more contemporary perspective, dengue has spread “rapidly” in the past 20 years, enabled by “increased global travel and the effects of climate change”. Between 2000 and 2019, WHO documented a “tenfold surge” in reported cases, to 5.2 million. Since then, the surge has continued; over 12.3 million cases were reported by the end of August 2024.
The global prevalence and effects of arboviruses like dengue are a “significant threat to public health”, particularly in tropical areas where they are endemic. Addressing this threat demands a “concerted, strategic, and informed response”, which the Director-General hopes to achieve with the SPRP, a “comprehensive plan” to outline ways of controlling Aedes-borne arbovirus transmission in affected countries.
“Our multifaceted approach emphasises integrated surveillance, laboratory diagnosis, vector control, community engagement, clinical management, and research and development.”
This approach should reduce the burden of disease, save lives, and minimise the socioeconomic consequences of these diseases. Furthermore, the Plan includes measures for “safe programming” to ensure interventions are “secure and do not exacerbate the risk” for those who are already vulnerable to disease or those involved in responding to the crisis. Dr Tedros states that prevention and control is a “shared responsibility”.
“Together, we can turn the tide against this disease, protect vulnerable populations, and pave the way for a healthier future.”
Understanding the threat
Dengue is a challenge across all of WHO’s regions, endemic in more than 100 countries. Various factors, such as unplanned urbanisation and the effects of climate change, fuel the spread of dengue and other Aedes-borne arboviruses, such as Zika and chikungunya, putting more than four billion people at risk. The growing threat must be addressed with a “robust and dynamic strategy” that accounts for the current global epidemiological landscape. This is complicated by the “still developing” global surveillance system.
Transmission drivers like the effects of climate change and population growth can explain the increase of these infections in some areas, but they also point to the need for a multisectoral approach to prevent and respond to outbreaks.
The Plan
The Plan is intended to “reduce the burden of disease and deaths from dengue and other Aedes-borne arbovirus diseases in all affected WHO regions”. The strategic objective is “to accelerate progress in preventing and controlling dengue and other Aedes-borne arboviral disease outbreaks worldwide”, with the following specific objectives:
- Strengthen global multisectoral coordination and collaboration among stakeholders and partners in preparedness, response, and resilience to dengue and other Aedes-borne arbovirus diseases
- Enhance the capacity of Member States in early detection, reporting, confirmation, and response to outbreaks of dengue and other Aedes-borne arboviruses
- Strengthen the capacity of Member States to implement effective vaccination and integrated vector management strategies for mitigating the transmission of dengue and other Aedes-borne arboviruses
The SPRP combines strategic interventions tailored to local contexts and leverages inter-stakeholder synergies to “confront the challenges” posed by these diseases and move closer to controlling them. The following “interconnected pillars” are included in the multidisciplinary approach:
- Leadership, coordination, planning, monitoring, and prevention of sexual misconduct
- Risk communication and community engagement (RCCE) and infodemic management
- Surveillance, case investigation, and contact tracing
- Travel, trade, and points of entry surveillance and control
- Laboratory and diagnostics
- Integrated vector management and WASH & IPC
- Clinical management and therapeutics
- Operational support and logistics
- Essential health services and systems
- Vaccination
- Research, innovation, and evidence
The 5Cs
The SPRP aligns with WHO’s 2023 Framework for Health Emergency Prevention, Preparedness, Response, and Resilience (HEPR) with a focus on five “core health emergency components”:
- Collaborative surveillance
- Strong national integrated disease, threat, and vulnerability surveillance,
- Effective diagnostics and laboratory capacity for pathogen and genomic surveillance
- Collaborative approaches for event detection, risk assessment, and response monitoring
- Community protection
- Community engagement, risk communication, and infodemic management
- Population and environmental public health interventions
- Multisectoral action for social and economic protection
- Access to countermeasures
- Fast tracked research and development
- Scalable manufacturing platforms
- Coordinated supply chains and emergency
- Emergency coordination
- Strengthened workforce capacity for health emergencies
- Strengthening health emergency preparedness, readiness, and resilience
- Health emergency alert and response coordination
- Safe and scalable care
- Scalable clinical care during emergencies
- Protection of health workers and patients
- Maintenance of essential health services
How do you think the SPRP can be effectively translated into specific contexts and implemented sustainably?
For insights into vaccination efforts for diseases that are being exacerbated by the effects of climate change, get your tickets to the Congress in Barcelona this month, or subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Oct 3, 2024 | Infection |
Since this article was published, Politico has reported that the previously suspected cases in Hamburg tested negative for Marburg virus with a PCR test and will be monitored throughout the incubation period of up to 21 days.
“There was no danger to fellow passengers on the train or in the plane at any time.”
(more…)

by Charlotte Kilpatrick | Sep 30, 2024 | Global Health |
Gavi announced two major funding updates at the United Nations General Assembly High-level week 2024, revealing that it is making progress in its fundraising efforts for the upcoming strategic period. The first of these updates is that the European Commission has pledged funding for the first two years of Gavi 6.0, complementing “strong support” from Team Europe and contributing to Gavi’s goal of helping to protect 500 million more children around the world. Gavi also announced an expanded collaboration with the United States International Development Finance Corporation (DFC), focussed on donor liquidity.
European support
The President of the European Commission, Ursula von der Leyen, addressed a crowd at the Global Citizen Festival on Saturday 28th September, revealing a funding pledge of €260 million for 2026-2027 and promising more to follow. The funds will support Gavi’s 2030 ambition of providing protection to 500 million more children, strengthening immunisation systems, and boosting global health security by “increasing readiness to respond to disease outbreaks”.
Added to the money pledged so far by the United States, France, Spain, and others in June 2024, this pledge takes Gavi’s total for the next strategic period to US$2.7 billion. The target is at least US$9 billion, which would enable Gavi to protect more children against more diseases, faster, and protect the world from outbreaks of disease when they occur. The €260 million pledge is for the first two years of Gavi’s upcoming strategic cycle, which coincide with the last two years of the EU’s 2021-2027 Multiannual Financial Framework (MFF). The European Commission is expected to remain committed to a “high level of ambition in supporting Gavi” as it prepares for the next MFF.
President von der Leyen reflected that “a healthier world is a better world”, with vaccination “one of our best chances for this”.
“Right now, millions of children are still at risk. We must continue to support vaccination around the world to save lives. So today I am proud to pledge 260 million euros for Gavi, the Vaccine Alliance. And more will come.”
DFC collaboration
The DFC and Gavi will expand their partnership with a focus on donor liquidity. This builds on support established during COVID-19, with the US$1 billion Rapid Financing Facility allowing Gavi to access funds quickly in the event of new donor pledges for pandemic response or routine immunisation. The mechanism is also central to Gavi’s Day Zero Financing Facility.
Nisha Biswal, DFC Deputy CEO, recognised that “global health security is economic and national security”. DFC invests in healthcare services, supply chains, and technology to strengthen pandemic preparedness and health system resilience, including over US$3 billion in health-related projects to enable over 50 million patients access healthcare.
“With the new Surge Financing Initiative, the expanded Gavi liquidity facility, and investments in regional manufacturing, we will be able to do far more to expand access to life-saving healthcare products, especially during health emergencies.”
Still on track
Dr Sania Nishtar, CEO of Gavi, expressed gratitude to the European Commission, recognising President von der Leyen’s “leadership in advancing global health outcomes” and DFC.
“Thanks to the European Commission and DFC, we remain on track to meet our target of protecting people, communities, even our entire world through immunisation.”
For more on global health investments at the Congress in Barcelona next month, get your tickets to join us here. Don’t forget to subscribe to our weekly newsletters for the latest vaccine news.

by Charlotte Kilpatrick | Sep 30, 2024 | Infection |
In September 2024, WHO’s Regional Office for Africa announced that Rwandan health authorities are “intensifying outbreak control efforts” after detecting Marburg virus disease in the country for the first time. 26 cases have been confirmed in seven of the country’s districts, and six deaths have been reported. The health authorities are implementing “comprehensive response measures” and an investigation to determine the origin of the outbreak. WHO states that it is supporting these efforts with expertise and tools.
Cases reported and response triggered
26 cases have been reported; 20 are in isolation and receiving treatment, and six deaths have been recorded. 161 contacts of the reported cases have been identified so far and are being monitored. The Ministry of Health, Rwanda, posted a video on social media with a caption reassuring viewers that “people can continue with their daily activities” and “should not panic” as the “hotspots of the disease” have been identified.
WHO is “mobilising” expertise and outbreak response tools to “reinforce the control measures” that are being rolled out. A consignment of clinical care and infection prevention and control supplies will be delivered from the WHO Emergency Response Hub in Nairobi, Kenya, to Kigali in the next few days. Efforts are also underway to “reinforce collaborative cross-border measures for readiness and response” in countries that neighbour Rwanda.
WHO Regional Director for Africa, Dr Matshidiso Moeti, explained that the critical outbreak response aspects are being put in motion “rapidly” to “halt the spread of this virus swiftly and effectively”.
“With the country’s already robust public health emergency response system, WHO is collaborating closely with the national authorities to provide the needed support to further enhance the ongoing efforts.”
Marburg
Marburg virus disease is a “highly virulent” member of the filoviridae family and causes haemorrhagic fever. It has a fatality ratio of up to 88%, with symptoms progressing rapidly after infection. The virus is transmitted to humans from fruit bats and spread among people through direct contact with the bodily fluids of infected people, surfaces, and materials.
The disease was first recognised after large outbreaks in Germany and Serbia in 1967, associated with laboratory research involving African green monkeys from Uganda. Outbreaks and cases have been reported sporadically since then, and efforts have been made to develop effective medical countermeasures. However, there is no licensed vaccine against Marburg virus disease.
A confluence of infectious disease
The Marburg outbreak will increase pressure on the Rwandan health system, which is already fighting its mpox outbreak, declared on 27th July 2024. In September 2024, Dr Jean Kaseya, Director-General of Africa CDC, reported that Rwanda had begun an mpox vaccine campaign after receipt of 1,000 doses. The campaign targeted districts bordering the Democratic Republic of the Congo, the epicentre of the current PHEIC. How will the health services respond effectively to both infectious disease threats, and will there be similarities or ‘doubling up’ in their strategies?
For the latest infectious disease and vaccine news, don’t forget to subscribe to our weekly newsletters here. To participate in important discussions about vaccine development in outbreak situations, join your colleagues at the Congress in Barcelona next month.

by Charlotte Kilpatrick | Sep 27, 2024 | Global Health |
A study in The Lancet Public Health in September 2024 evaluates the measles dynamics in England between 2010 and 2019 to understand the effects of waning of vaccine-induced immunity. The researchers find that, although the MMR vaccine remains “highly protective” against measles infections for decades, and most transmission is “connected to people who are unvaccinated”, breakthrough infections in vaccinated individuals aged 15 years or older are “increasingly frequent”. However, they emphasise the importance of adequate coverage alongside vaccine effectiveness.
In England, measles “follows typical near-elimination transmission dynamics”, with “sporadic localised outbreaks and high national vaccine coverage”. England reached measles elimination status after “large outbreaks” between 2011 and 2013. From 2017 onwards a resurgence has been observed.
Highly protective vaccines
The authors describe measles vaccines as “highly protective against infection” recognising that they enabled a “great decrease in the global burden of measles” after immunisation programmes began in the 1970s and 1980s. Indeed, some countries became eligible for an elimination status since 2000 after the successful implementation of routine immunisation programmes. However, this is slipping out of reach for many countries in Europe and the Americas, which have reported a resurgence between 2015 and 2020.
“This resurgence was mostly reported in under-immunised communities and linked to past variations in vaccine coverage.”
Further outbreaks have been reported in “highly vaccinated” groups in Portugal and Japan, inviting questions about the waning of measles immunity in adults who had received two doses in childhood. Research suggests a waning of antibodies in young adults who had received two doses of vaccine “more than 20 years earlier”, in contrast to no decrease in previously infected individuals. Analysis of outbreak data suggest a “drop” in vaccine effectiveness in young adults who had received two doses of vaccine. However, effectiveness estimates appear to be “sensitive to assumptions on infection-induced immunity”.
The study
The study addressed the need to understand whether the measles case dynamics of settings with high vaccine coverage result from a waning of vaccine-induced immunity or if changes in the distribution of immunity in the population are driving the distribution of vaccine status among cases. A mathematical transmission model, stratified by age, region, and vaccine status was used to evaluate whether the measles dynamics in England from 2010 to 2019 were “in line with a waning of vaccine-induced immunity”. Three scenarios were modelled:
- Vaccinated individuals might only become infected because of primary vaccine failure
- Vaccinated individuals might become infected because of primary or secondary vaccine failure, with the risk of secondary vaccine failure depending on age
- Vaccinated individuals might become infected because of primary or secondary vaccine failure, with the risk of secondary vaccine failure depending on age and time since measles stopped being endemic
Each scenario was fitted to measles case data reported in England between 2010 and 2019, and the authors compared the resulting performance. Data were collected by UKHSA (formerly Public Health England), and included date of symptom onset, region of residence, age, and vaccine status. The final case dataset included 7,504 cases. The annual proportion of individuals who had been infected with measles and received two doses of the vaccine out of the overall number of individuals with measles was three times higher in 2019 than in 2011. The median age of individuals with measles was 12.5 years.
Results
Scenarios integrating waning of vaccine-induced immunity “better captured measles case dynamics” than the scenario without waning. In the scenario where waning started in 2000, the estimated waning rate was 0.039% per year.
“Although slow, waning was associated with an increased burden over time; setting the waning variable in this scenario to 0 led to a substantial decrease in cases.”
While overall vaccine effectiveness was estimated to stay high over the decades, the estimation suggested that the increasing number of breakthrough infections contributed to the measles burden in England. The additional burden brought by waning is “directly related to the risk of transmission from vaccinated cases”, as individuals infected by people who had been vaccinated would not have otherwise been infected.
“Our results suggest that the waning of vaccine-induced immunity likely explains the observed dynamics and age distribution of vaccinated measles cases in England between 2010 and 2019.”
Low vaccination rates a bigger factor
Dr Alexis Robert, Research Fellow in Infectious Disease Modelling at London School of Hygiene and Tropical Medicine (LSHTM) drew attention to the “biggest factor for measles outbreaks”: low vaccination rates. Dr Robert emphasised that the MMR vaccine is “highly effective” and two doses “will protect you and those around you”.
“This 0.04% waning each year is relatively slow, but because measles is so infectious, over time, this would add up to a ‘gap’ in a population’s defences the virus can exploit, which may increase the duration and size of outbreaks.”
The data patterns in the study emerge “because outbreaks have occurred as a result of declines in vaccine coverage”, said Dr Robert.
“If there were no outbreaks, this small amount of waning would not show up in any data. The key issue here is coverage, not the effectiveness of the vaccine.”
Dr Anne Suffel, co-author from LSHTM, agreed that the study “looks at one small part of the picture” and recognised that the “larger issue” is that “uptake of the MMR vaccine has been decreasing in England since 2015”.
“Understanding the impact of vaccine immunity waning will help anticipate the potential impact of measles in countries where incidence has been low for decades, but vaccine uptake is reducing. The best way to limit the impact of measles and protect everyone from what can be a horrible disease, is to keep vaccine uptake as high as possible.”
Dr Adam Kucharski, Professor of Infectious Disease Epidemiology and co-author from LSHTM, acknowledged the role of “other factors” such as “changes in testing patterns over time”.
“However, the consistency and age distribution of the increase in England – combined with reports of cases in vaccinated individuals in other countries and previous laboratory studies showing a decline in measles antibodies – suggests a biological explanation is involved.”
Join us at the Congress in Barcelona next month to explore the reasons for a resurgence in measles from an uptake perspective, and don’t forget to subscribe to our weekly newsletters for more vaccine news.

by Charlotte Kilpatrick | Sep 20, 2024 | Global Health |
In September 2024, the European Medicines Agency (EMA) announced its recommendation to extend the indication of the smallpox and mpox vaccine Imvanex to adolescents from 12 to 17 years of age. Imvanex, Bavarian Nordic’s MVA-BN, is authorised in the EU for the protection of adults against smallpox, mpox, and disease caused by the vaccinia virus. The Committee for Medicinal Product for Human Use (CHMP) based this recommendation on interim results from a study comparing the effects of the vaccine in adolescents and adults.
Data-led expansion
Interim results indicate that the immune response in adolescents was comparable to adults, from which the authorities have inferred that the vaccine will “provide similar protection in adolescents to that expected in adults”. The safety profile was also comparable with no additional risk identified. EMA has requested the marketing authorisation holder to submit the results of this study by 30th May 2025 to further characterise the safety information for adolescents.
Although this is the first approval of MVA-BN as a smallpox,mpox vaccine for adolescents, Bavarian Nordic notes that a recombinant version of MVA-BN (Mvabea) received EMA approval in 2020 as part of a prime-boost regimen for the prevention of disease caused by Ebola virus in individuals aged 1 and older. This approval was based on studies involving more than 3,300 individuals, including over 800 children and adolescents aged 1-17 in Africa.
Implications for the response
EMA states that this assessment has “important implications” for the global mpox response. As EMA is the regulatory agency of record for the WHO prequalification of the vaccine earlier this month, the CHMP assessment constitutes the basis for WHO prequalification approval to “facilitate timely and increased access” in communities that need it most. The EMA’s assessment has also previously been considered by the DRC’s national regulatory authority for fast-track approval.
President and CEO of Bavarian Nordic, Paul Chaplin, applauded EMA for the “expedited” review and decision.
“This represents an important milestone in our efforts to make our vaccine available for all populations and will help improve access for some of the most vulnerable individuals mostly impacted by the ongoing mpox outbreak in Africa.”
For insights into the regulatory processes behind access to vaccines, including from senior representatives of EMA, join us at the Congress in Barcelona next month. Don’t forget to subscribe to our weekly newsletters for the latest vaccines news.

by Charlotte Kilpatrick | Sep 20, 2024 | Global Health |
In September 2024, Africa CDC offered congratulations to the Governments of Japan and the Democratic Republic of the Congo (DRC) for their agreement on the donation of Lc16 mpox vaccines and specialised inoculation needles. At a signing ceremony in Kinshasa, the two governments marked a “significant milestone” in their cooperation. These vaccines are a “critical boost” to DRC’s mpox response; Lc16 is the only mpox vaccine currently approved for children.
Lc16
The vaccine is derived from the Lister strain of vaccinia and contrasts to the replication-deficient vaccines like Modified Vaccine Ankara (MVA) by retaining most of vaccinia genome and being able to replicate at the site of inoculation. It is administered as a single dose through the scarification method, which involves scratching the skin before applying the vaccine solution. Studies in children suggested that the vaccine was safe and well tolerated, leading to licensure. However, the duration of immunity remains unclear.
Another tool for the fight
Africa CDC states that this donation comes “at a pivotal time” in DRC’s fight against the regional mpox outbreak. Since the declaration of a PHECS in August 2024, a “more coordinated international response” has evolved. Central to this response is vaccination, which is complicated by Africa’s “limited access to critical countermeasures”.
Director General of Africa CDC, H.E. Dr Jean Kaseya, is “deeply appreciative” of the ‘” of the “generous donation” of mpox vaccines and specialised vaccine needles to the DRC.
“This timely assistance will significantly bolster our ongoing efforts to contain the outbreak, and I am confident that this partnership will help mitigate the public health threat posed by mpox, not only in the DRC but across the continent.”
Japan’s Senior Deputy Minister for Foreign Affairs, Takeshi Akahori notes the increasing number of cases in the DRC and other countries, commenting that Japan is “monitoring the situation closely” with WHO.
“I hope that these vaccines and needles will contribute meaningfully to the fight against mpox.”
Join us at the Congress in Barcelona next month to engage with public health experts on current health threats and how vaccines can contribute to our response, and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 18, 2024 | Global Health |
The Global Fund to Fight AIDS, Tuberculosis, and Malaria (the Global Fund) announced in September 2024 that it is responding to a request from the government of the Democratic Republic of the Congo (DRC) for support in its mpox response. The Global Fund will provide US$9.5 million towards the emergency response in six of the highest transmission provinces (Equateur, Sud-Ubangui, Sankuru, Tshopo, Sud-Kivu, and Nord-Kivu) as well as Kinshasa, which is home to 17 million people.
DRC is fighting the world’s largest mpox epidemic, reporting 5,160 confirmed cases and 25 deaths since the beginning of the year. However, as testing capacity and availability are limited, the number of suspected cases could be up to five times the number of laboratory-confirmed cases. As the epidemiology of mpox evolves in “complex ways”, implications for prevention, preparedness, and response efforts also evolve. Furthermore, mpox is “increasingly being associated with HIV”, which heightens the risk of transmission, illness, and death. Thus, investment in mpox efforts helps to strengthen work on HIV, and vice versa.
Global Fund support
The support offered by the Global Fund contributes to specific priority areas on the government’s National Preparedness and Response plan:
- Enhancing disease surveillance systems with an emphasis on strengthening early warning capabilities and alert and response systems to detect, monitor, and respond to mpox and other disease outbreaks.
- Strengthening laboratory systems and diagnostics to increase case detection and stop the spread.
- Conducting risk communication and community mobilisation and engagement through the network of community health workers and community actors who are deployed for HIV, TB, and malaria prevention and awareness.
- Implementing infection prevention and control measures to protect health workers, including at the community level.
- Reinforcing country-level coordination, planning, and support for emergency response and operations.
- Strengthening the capacity of health facilities to provide primary care services and support future emergencies.
The support also complements the current collaboration between DRC’s ministry of health, Africa CDC, WHO, humanitarian organisations, and other key partners on efforts to “address the severe challenges to the public health system” in the east of the country, where the epidemic is “converging with risks of other infectious diseases”. The Global Fund has already contributed support in moving available stocks of personal protective equipment to the most affected provinces.
Today’s fight for tomorrow’s resilience
Dr Roger Kamba, Minister of Health and Social Welfare for the DRC recalled the “proven track record” of infectious disease control in the partnerships with the Global Fund and other health partners.
“Over the past two decades, the number of AIDS-related deaths and new HIV infections in DRC have reduced by more than 60%, through coordination and collaboration across all out partners.”
Dr Kamba is “determined to continue to work in the same manner for a strong response to mpox”.
“The fight against the current mpox epidemic is a top priority for our ministry, especially through the reinforcement of the community response. It is essential to recognise that by acting now, we are not only fighting mpox but also investing in the resilience and health security of tomorrow.”
Executive Director of the Global Fund, Peter Sands, reflected that people who live in “areas of conflict and crises” often encounter “significant barriers to accessing health services”.
“When a disease outbreak occurs in these places, the challenges are compounded. Strong systems of trusted community health workers, health educators, and other local responders are essential for stopping disease spread.”
Mark Edington, Head of Grant Management at the Global Fund, emphasised the importance of swift action in disease outbreak situations.
“Immediate intervention is crucial to strengthen systems for health and improve disease detection, surveillance, and response mechanisms, aiming to prevent further deterioration in health outcomes, particularly for women, children, and internally displaced persons.”
The Global Fund encourages other affected countries to assess their mpox needs and the consider the possibility of repurposing existing Global Fund investments.
To contribute to important discussions about effective allocation of funds for emergency responses and ensuring more resilient health systems, join us at the Congress in Barcelona next month. Don’t forget to subscribe to our newsletters here for the latest vaccine and global health news.

by Charlotte Kilpatrick | Sep 16, 2024 | Global Health |
In September 2024 WHO announced the establishment of an access and allocation mechanism for mpox medical countermeasures, including vaccines, treatments, and diagnostic tests. The Access and Allocation Mechanism (AAM) is intended to increase access to these essential tools for people at highest risk, ensuring that limited supplies are used “effectively and equitably”. This announcement comes after WHO declared the mpox outbreak a PHEIC in August 2024 and addresses one of the key International Health Regulations Emergency Committee’s recommendations: “equitable access to safe, effective, and quality-assured countermeasures”.
AAM
The AAM is part of the interim Medical Countermeasures Network (i-MCM-Net). Developed in response to “global vulnerabilities” exposed by the COVID-19 pandemic, i-MCM-Net enhances collaboration through a “Network of Networks” approach. It seeks to provide timely and equitable access to quality, safe, effective, and affordable medical countermeasures in response to public health emergencies through existing networks and global collaboration. The network was endorsed by WHO Member States as an interim mechanism while negotiations on a pandemic agreement continue.
The mpox AAM includes members of the i-MCM-Net as well as WHO: Africa CDC, CEPI, the EU Health Emergency Preparedness and Response Authority (HERA), FIND, Gavi, the PAHO Revolving Fund, UNICEF, Unitaid, and others. It will work to allocate the “currently scarce supplies” to those at highest risk of infection.
It will operate according to three guiding principles:
- Preventing illness and death – prioritise vaccination and other tools to interrupt transmission for those at greatest risk to prevent illness and death.
- Mitigating inequity – ensure equitable access to medical countermeasures for all people at risk, irrespective of socio-economic or demographic background.
- Ensuring transparency and flexibility – establish and maintain clear and open communication about allocation decisions and be ready to adapt strategies as new data emerge or situations change.
More than 3.6 million vaccine doses have been pledged for the mpox response, including 620,000 doses of MVA-BN pledged to affected countries by the European Commission, Austria, Belgium, Croatia, Cyprus, France, Germany, Luxembourg, Malta, Poland, Spain, and the United States of America, as well as Bavarian Nordic. Japan has pledged 3 million doses of the LC16 vaccine. This is the largest pledge so far.
International coordination
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, recognised the need for “powerful tools” like vaccines, therapeutics, and diagnostics, to bring the mpox outbreak “under control”.
“The COVID-19 pandemic illustrated the need for international coordination to promote equitable access to these tools so they can be used most effectively where they are most needed. We urge countries with supplies of vaccines and other products to come forward with donations, to prevent infections, stop transmission, and save lives.”
Dr Mike Ryan, Executive Director of WHO’s Health Emergencies Programme, emphasised that WHO and its partners are working with the government of the Democratic Republic of the Congo and other affected countries to “implement an integrated approach to case detection, contact tracing, targeted vaccination, clinical and home care, infection prevention and control, community engagement and mobilisation, and specialised logistical support”.
“The AAM will provide a reliable pipeline of vaccines and other tools in order to ensure the success on the ground in interrupting transmission and reducing suffering.”
Join us at the Congress in Barcelona next month to share your insights on the best ways to ensure equitable access to essential medical countermeasures, and don’t forget to subscribe to our weekly newsletters for the latest vaccine news.

by Charlotte Kilpatrick | Sep 13, 2024 | Technology |
In September 2024, WHO announced that the MVA-BN vaccine, manufactured by Bavarian Nordic, is the first vaccine against mpox to be put on the prequalification list. This approval is “expected to facilitate timely and increased access” to the vaccine in “communities with urgent need” amid the outbreak of mpox. The prequalification is based on information submitted by Bavarian Nordic and reviewed by the European Medicines Agency.
Recommended for use
WHO’s Strategic Advisory Group of Experts (SAGE) on Immunisation recommended the use of MVA-BN in an mpox outbreak context for persons at high risk of exposure. It can be administered in over-18s in two doses 4 weeks apart. After initial cold storage it can be kept between 2°C and 8°C for up to 8 weeks.
Although it is not currently licensed for persons under 18 years of age, the vaccine can be used “off-label” in infants, children, and adolescents, and in pregnant and immunocompromised people. This allows use in an outbreak after consideration of the potential risks in relation to the benefits of vaccination.
Data available to WHO reveal that a single-dose MVA-BN vaccine, administered before exposure, has an estimated 76% effectiveness in protecting against mpox; the two-dose schedule achieves an estimated 82% effectiveness. Post-exposure vaccination is less effective. Clinical studies have demonstrated a good safety profile and vaccine performance, which have been confirmed in real-world use during the global outbreak since 2022. However, WHO emphasises the need to collect “as much data as possible on vaccine safety and effectiveness in different contexts”.
An important step
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, described this first prequalification as an “important step in our fight against the disease” with implications for current outbreaks and the future.
“We now need urgent scale up in procurement, donations, and rollout to ensure equitable access to vaccines where they are needed most, alongside other public health tools, to prevent infections, stop transmission, and save lives.”
WHO Assistant Director-General for Access to Medicines and Health Products, Dr Yukiko Nakatani, suggested that the prequalification will “help accelerate ongoing procurement” to help communities “on the frontlines of the ongoing emergency in Africa and beyond”.
“The decision can also help national regulatory authorities to fast-track approvals, ultimately increasing access to quality-assured mpox vaccine products.”
Dr Rogerio Gaspar, WHO Director for Regulation and Prequalification, commented that the findings of the product and programmatic suitability assessments are “particularly relevant in the context of the declaration of a public health emergency of international concern (PHEIC)”.
“We are progressing with prequalification and emergency use listing procedures with manufacturers of two other mpox vaccines: LC-16 and ACAM2000. We have also received 6 expressions of interest for mpox diagnostic products for emergency use listing so far.”
To explore the steps before, during, and after these approval processes in emergency contexts, join us at the Congress in Barcelona this October. Don’t forget to subscribe to our weekly newsletters here for regular updates.

by Charlotte Kilpatrick | Sep 12, 2024 | Global Health |
Bavarian Nordic issued an update on its supply and manufacturing activities in support of mpox response efforts in September 2024. The company manufacturers the approved non-replicating smallpox and mpox vaccine MVA-BN (marketed as JYNNEOS, IMVAMUNE, and IMVANEX). The company statement describes “intensified” collaboration with global stakeholders in response to the declaration of mpox as a PHECS by Africa CDC and a PHEIC by WHO.
Progress so far
Through donations from the European Commission, the United States government, and Bavarian Nordic, initial doses of MVA-BN have been delivered to the Democratic Republic of the Congo (DRC), the epicentre of the mpox outbreak. Further donations have been pledged by other countries. However, UNICEF and Africa CDC have suggested that vaccines will be needed to protect up to 1 million people in high-risk areas of DRC, with up to 10-12 million doses required through 2025.
Bavarian Nordic “is committed to ensuring equitable access to its mpox vaccine”. It has therefore prioritised the production of MVA-BN for the rest of the year to ensure up to 2 million doses by year-end. This means that some existing orders for 2024 will be delayed to 2025 to provide “greater flexibility” to meet “additional urgent and imminent needs”. The new supply contracts include multi-year agreements with countries, including those who have pledged vaccines for Africa. Additionally, Bavarian Nordic has responded to UNICEF’s emergency tender and is participating in discussions with other organisations and individual governments around the globe.
Up to 13 million doses
By focussing “full capacity to address the current public health emergency”, Bavarian Nordic hopes to supply up to 13 million MVA-BN doses by the end of 2025. This includes 2 million in 2024. Although this may be sufficient to meet the current and near future demand, Bavarian Nordic is exploring “additional levers” to expand capacity. This includes possibly transferring manufacturing to other companies.
“Based on these early, but highly constructive discussions, together with further planned improvements in the manufacturing process, Bavarian Nordic has identified another 50 million doses that, pending regulatory approvals and demand, could be supplied during the next 12-18 months.”
President and CEO of Bavarian Nordic, Paul Chaplin, stated that the company is “working closely with all governments and organisations to support the international efforts to combat the latest public health emergency”. Mr Chaplin highlighted that Bavarian Nordic will “support all requests for vaccine and have already secured agreements to the UNICEF tender that will hopefully secure more access to MVA-BN globally”.
“We remain committed to the equitable access either through prioritising our own capacity, accelerating planned improvements in the manufacturing process and by exploring ways to further expand capacity through partnerships around the globe. Once again Bavarian Nordic, through innovation and our commitment to improving and saving lives has stepped forward as an important part of the international community’s response to the current public health emergency.”
We will hear more from Bavarian Nordic on their contributions to the mpox response at the Congress in Washington next April; get your tickets to join us there and don’t forget to subscribe to our weekly newsletters here.

by Charlotte Kilpatrick | Sep 11, 2024 | Global Health |
Gavi announced in September 2024 that a shipment of 15,460 doses of mpox vaccine has arrived in the Democratic Republic of the Congo (DRC) to “support the global and regional effort to contain the outbreak”. These doses were donated by manufacturer Bavarian Nordic to Gavi-eligible countries. Arriving in Kinshasa, the shipment adds to the 215,000 vaccine doses already donated by the European Union.
Global response
The DRC is the epicentre of the mpox outbreak; it has reported around 94% of all cases and 99% of related deaths. It has secured national regulatory approval of Bavarian Nordic’s JYNNEOS mpox vaccine and rollout is expected to begin shortly. Gavi has made US$2.9 million under the Fragility, Emergencies, and Displaced Populations (FED) Policy to support DRC’s vaccination efforts.
In August 2024, Gavi announced that the Board has approved the final terms of the First Response Fund. This is the “fastest tool in a suite of instruments” known as the Day Zero Financing Facility; this is intended to make resources “immediately” available for a vaccine response to an urgent public health emergency. Gavi is also working with countries and partners and sharing “legal and process knowledge” and operational support to Africa CDC and partners.
“Based on our experience working with donors, countries, and partners to coordinate the global COVAX dose donation mechanism, this includes providing information on the complex technical, legal, regulatory, and logistical considerations involved in mounting rapid vaccination campaigns with donated doses.”
CEO of Gavi, Dr Sania Nishtar, described the latest shipment as a “timely addition” to the global response.
“Now that they have arrived, our first priority is to work with our partners to ensure these and other vaccines reach those who need them, as quickly and efficiently as possible. Gavi has already unlocked funds to support this delivery.”
For regular updates on the global effort to stop the spread of mpox with vaccines, don’t forget to subscribe to our weekly newsletters here. To engage with experts working in this area, get your tickets to the Congress in Barcelona this October.

by Charlotte Kilpatrick | Sep 5, 2024 | Global Health |
Bavarian Nordic announced in September 2024 that the first doses of its mpox vaccine, MVA-BN (JYNNEOS), have arrived in the Democratic Republic of Congo (DRC) to support response efforts. Further shipments are planned within the next few days, contributing to a total of more than 250,000 doses donated by the European Commission’s Health Emergency Preparedness and Response Authority (HERA), the United States government, and Bavarian Nordic.
First doses arrive
This shipment brings the first mpox vaccines to the DRC, which Bavarian Nordic describes as a “turning point” in the joint efforts by Africa CDC and the international community. The DRC authorities have issued national emergency use authorisation for MVA-BN to allow immediate deployment. Broader deployment on the continent is “pending an emergency use listing” from WHO.
WHO Director-General Dr Tedros Adhanom Ghebreyesus shared his reaction to the delivery on social media, emphasising that WHO and partners are supporting DRC in the delivery of a “comprehensive health emergency response based on case finding, contact tracing, targeted vaccination, case management, and community engagement and mobilisation”.
“Once again, we are grateful to the European Union for its solidarity and for sharing vaccines.”
Responding to the call
DRC is the “epicentre” of the African mpox outbreak, reporting more than 94% of reported cases and nearly 99% of the reported deaths year-to-date on the continent. Although surveillance and control are “continuously improving” the true burden of disease remains unclear as case numbers are “underreported”. Bavarian Nordic’s President and Chief Executive Officer, Paul Chaplin commented on the “alarming rate” of increased cases in the DRC, where “aid is desperately needed”.
“Africa CDC has called for the international community to step up and mobilise the resources needed to combat the outbreak, and we are proudly responding to this call together with our partners in the European Commission’s Health Emergency Preparedness and Response Authority and the U.S. government, who have demonstrated strong leadership and determination during this serious health crisis.”
Mr Chaplin stated that “time is of the essence” but is pleased that “the way has now been paved for our mpox vaccines to reach the people in the DRC who are most in need”.
“We will continue our collaborative efforts with both international partners and local authorities to further broaden access to our vaccine in the region.”
While this news is welcomed by public health experts, will the doses be enough to control the outbreak, and is there a risk of perpetuating dependence on donations? For more on preparedness and response efforts at the Congress in Barcelona this October, get your tickets now. Don’t forget to subscribe to our weekly newsletters here.